Since the late nineteenth century, researchers have explored a stream of derivatives within the pyrazolone family. The main idea was to improve options in pharmaceuticals, dyes, and analytical chemistry. Over time, a handful of pyrazolone derivatives earned reputations for strong analgesic and anti-inflammatory activity. Chemists in the mid-twentieth century started steering attention toward halogen-substituted versions, picking up on altered reactivity and biological behavior. Against this backdrop, 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone didn’t rise to fame overnight. Its development owes a lot to advances in organic synthesis, persistent curiosity about tweaking molecular frameworks, and an eye for applications outside straightforward drug development. Every new substitution pattern represented more possibility. Looking through old literature, I noticed that interest in these kinds of molecules always seemed to surge with each new analytical method or pharmacological discovery.
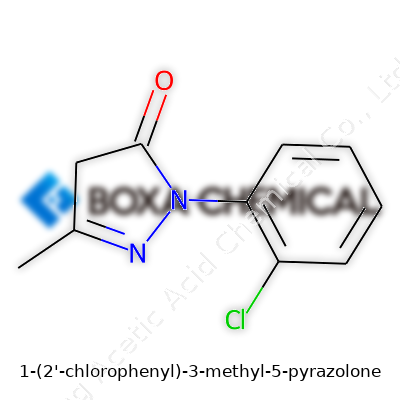
At core, 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone is a heterocyclic compound built on the five-membered pyrazolone ring. It carries a chlorine-tagged phenyl group in the 1-position and a methyl group at the 3-position. This setup steers both physical traits and how it reacts with other chemicals. In pharmaceutical and analytical work, this specific arrangement speaks to a push for better potency, solubility, and performance. Researchers turn to such tailored molecules when standard options start to fall short in selectivity or response time, or when they need something more robust for industrial assays or intermediate steps.
Many pyrazolones, especially those tweaked with halogens like chlorine, show up as crystalline powders with stark melting points and solid thermal stability. The introduction of a 2-chlorophenyl group usually leads to improved resistance against oxidation and a certain sturdiness under normal laboratory conditions. Solubility often falls between moderate and low in water, but goes up in organic solvents—an asset during extraction or chromatography. Having handled compounds with this backbone, I pay attention to their aromatic odor, the way they crystalize in small, prismatic grains, and their reluctance to dissolve in plain water. These characteristics matter not just for handling but for end-use: stability and solubility often define how a molecule gets used in pharmaceuticals or dye manufacture.
On the market, 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone usually carries clear, concise labeling: chemical name, structural formula, purity (often upwards of 98 percent for research and synthesis), batch data, molecular weight, and sometimes UV/Vis spectral data. Labs checking for compound integrity typically expect strong compliance with IUPAC naming, clear hazard pictograms, CAS numbers, and storage conditions—cool, dry, and away from direct sunlight. For someone in quality control or research, proper labeling and documentation provide assurance against costly mix-ups. Clear technical specs help trace molecular performance back to batch data whenever odd results pop up in downstream testing or production.
Most syntheses for this compound start with condensation of a 2-chlorophenylhydrazine with ethyl acetoacetate or a related diketone under mild acid or base catalysis. Temperature control steers cyclization, and isolation usually goes through recrystallization with organic solvents. Having run parallel condensations myself, it’s easy to see why simple, direct routes stay popular in both industry and academia—lower cost, higher yield, and fewer purification headaches. Tweaks in choice of solvent, base, or temperature can mean the difference between an efficient, repeatable reaction and frustrating waste. Reliability in the preparation method matters for both safety and reproducibility, especially once pilot batches move to larger-scale runs.
The backbone of 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone offers lots of room for creativity. The chlorine atom at the ortho-position readies this compound for nucleophilic substitution, letting chemists tack on new side chains or groups and widen its application. The methyl at the 3-position stays put, but the carbonyl and pyrazolone ring provide anchors for further derivatization. In the lab, these features open the door to forming metal complexes, engaging in azo coupling, and exploring structure-activity relationships in medicinal chemistry. Anyone trying to optimize physical or pharmacological profiles focuses on these handles. Each step away from the parent compound could mean new activity, less toxicity, or better performance in technical applications.
Scientific literature and catalogues describe this molecule using several names, often shifting between common naming conventions and IUPAC: 1-(2-chlorophenyl)-3-methyl-2,4-dioxo-1,2-dihydropyrazol-5-yl, or just the more casual paraphrase: 2-Chlorophenyl methyl pyrazolone. As chemists, we watch these permutations partly for clarity in ordering or searching literature, partly to avoid confusion in cross-regional collaboration. Sometimes trade names pop up in dye or pharma outputs, but at research scale, the full chemical name usually leads communication.
Halogenated pyrazolones carry their own set of risk profiles. Dust can irritate, and some derivatives show low-level toxicity even at modest exposures. Given the potential for allergic reactions or sensitization, most labs enforce skin and eye protection, fume hoods during synthesis, and containment during weighing or transfer. Anyone who’s handled solid, aromatic pyrazolones can attest: a small lapse in attention, a gust of powder, and it takes days to clear the odor. Safety data sheets urge avoiding skin contact and inhalation, keeping storage dry and away from oxidizers or acids. Proper disposal in line with institutional and local hazardous waste protocols rounds out responsible handling. Sticking with standardized personal protective equipment, open benches for dilution or measurement, and good chemical hygiene cuts risk for everyone on the team.
1-(2-Chlorophenyl)-3-methyl-5-pyrazolone shows up in analytical chemistry as a chelating agent and intermediate, in dye chemistry, and sometimes as a template for new pharmacophores. Its structural features help bind iron and other transition metals, making it practical for colorimetric assays in water analysis. Some researchers look to it and close relatives as possible anti-inflammatory or anti-microbial leads, especially as antibiotic resistance rises and the need for fresh analogs grows more urgent. In dye chemistry, the resonance and halogenation suit it for stable, vivid pigments, especially in environments where light fastness or chemical resistance matter. Colleagues in materials science occasionally test such molecules as stabilizers or functional additives in new coatings and polymer blends. The broad utility comes less from being a “wonder molecule” than from adaptability built into its chemistry.
Efforts to improve 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone focus on widening biological activity or improving performance in chemical detection. Research groups keep chipping away at modifications—swapping groups around the phenyl or pyrazolone ring, coupling to novel scaffolds, or tightening analytical sensitivity for assays. As published work in prominent journals shows, advanced spectroscopic and crystallographic tools now let teams pinpoint structure-activity relationships faster and with less guesswork than anyone had decades ago. The pace of new analogs and the depth of biological screens keeps rising, driven by the search for safer, more precise therapeutics or more reliable analytical reagents. Anyone who’s spent time at the bench knows the challenges of keeping up—every relevant tweak means another round of synthesis, characterization, and bioassay, pushing both cost and time.
Some derivatives in the pyrazolone family grab headlines for toxicity—think antipyrine, phenylbutazone, or certain dye intermediates. For 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone, scattered reports flag mild to moderate toxicity risk, with eye on metabolic breakdown and possible impact on liver and kidney function. My own reading of toxicology studies suggests that risk sits squarely with chronic exposure or high-concentration handling, less so with routine analytical use under controlled lab conditions. Regulatory agencies emphasize full risk assessments before new pharmaceutical or agrochemical applications get green-lighted. This is where ongoing research plays a vital role: every new data point sharpens our understanding of dose, route of exposure, and at-risk populations. Scientists in risk assessment urge attention to off-target effects, metabolites, and cumulative exposure, not just acute toxicity endpoints. Rigorous pre-clinical work, standard setting by local authorities, and open reporting of incident data give downstream users confidence or at least a clear sense of limits.
Looking forward, 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone stands as a stepping-stone for new hybrid molecules in pharmaceutical discovery and analytical chemistry. Its demonstrated reactivity and modifiable structure hold promise for advanced chelators, precision dyes, and possible anti-infectives as resistance patterns shift. I expect to see continued innovation merging old-school organic synthesis with computational prediction, speeding up discovery of analogs with better safety or stronger performance. Regulatory changes, increased demand for green chemistry, and tighter workplace safety protocols drive further scrutiny, underscoring the need for deeper toxicity and environmental impact data. While such compounds can never be “risk-free,” knowledge and respect for their chemistry will define their safest, most productive paths moving ahead.
Most people have never heard of 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone. For many, it sounds like just another chemical drifting around in the world of industrial science. But spend any time studying pharmaceuticals or advanced pigment technologies, and you start running into names like this. For those working in certain labs, it’s as familiar as aspirin on the shelf.
One of the most immediate places you’ll run into this substance is in the pigment industry. Back in my university days, I took a part-time job helping a friend with organic synthesis. Much of our work involved derivatives of pyrazolones, with 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone showing up as a key starting material. This compound often becomes a core building block for making pigments like Pigment Yellow 13 and similar dyes used in plastics, paints, and inks.
Scientists blend 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone with other chemistries, creating a range of colors with properties needed for tough jobs. Car finishes, for example, face years of sunlight, rain, and washing. Paint makers look for molecules that don’t fade or break down. This is where the compound’s reliability comes in handy. Pyrazolone-based pigments bring solid color to everything from road signs to packaging.
I once shadowed a pharmaceutical chemist. Shuffling index cards on his desk, he explained how small changes in a molecule could help designers find new drugs. While 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone isn’t a blockbuster painkiller itself, its structure matters. Molecules with similar frames have shown activity as analgesics or anti-inflammatories. Its skeleton crops up in research looking at new medicines, giving chemists flexibility to explore uncharted ground.
There’s another use, less publicized but just as critical. Medicinal chemistry always needs platforms for building more complex molecules. Many tweaks and tests begin with something sturdy like this—reactive but manageable, and a good place to start tailoring new compounds. Scientists take childhood lessons of mixing things together and gradually scale that up into discoveries that help fight disease.
Experience shapes respect for chemicals like this one. Colleagues developing new pigments or researching drug candidates know safety remains top priority. Working with materials that bring both opportunity and risk means keeping skill and knowledge at the center. Chemistry isn’t just about bright colors and laboratory breakthroughs. Health risks can run higher with some aromatic organics, and 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone fits that mold—personal protective gear, proper ventilation, and good housekeeping go hand in hand with discovery.
Awareness about ingredients hidden behind long chemical names matters. Manufacturers, researchers, and regulatory agencies all play a part in balancing innovation and safety. As industries search for more sustainable options, interest in the environmental footprint of chemical starting materials has grown. Building better transparency, both in supply chains and public knowledge, helps address concerns before they become problems.
From bright pigments in everyday products to scaffolding for future medicines, 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone proves how one compound can shape both art and science. Not everyone needs to know how to pronounce it, but understanding what it brings to the table—and how we manage its risks—moves everybody forward.
People who work in labs, factories, or even research desks connected remotely to supply lines all know one thing: mistakes with chemicals don’t care about degrees or experience. 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone isn’t the most famous name outside chemistry circles, but once you’re dealing with compounds even close to its class, the risks change the game.
In my own days shadowing experienced chemists and techs, I caught quiet lessons that books miss. Gloves stained or reused. A mask dropped to sip coffee. I’ve seen sharp minds let small habits slide—until one surprise reaction bench-side, or the headache that clings for hours too many. Far from an abstract danger, small lapses around chemicals with complicated names can open the door to long-term health problems or sudden emergencies.
Touching this compound, even with a trace on bare skin, spells trouble. Redness or peeling might start mild, but repeated handling builds a bigger risk story. Proper gloves—nitrile, not the cheap kind that split when catching on rough glass—cover hands, and eye protection blocks the fine dust or splash. Splash shields become more than an afterthought when even opening a bottle could send a breeze of powder on the air. It helps to keep a wash station primed, not boxed up for later.
Powder drifting on a small puff of wind or blown from a scooper hits more deeply than a faint chemical whiff. Inhaling 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone’s dust can leave the lungs vulnerable. Instead of assuming a lab’s fume hood always works, I learned to check airflow myself—hold a tissue at the edge and see it pull in. Even outside a high-tech lab, N95 respirators or similar face covers set a line between exposure and safety. Open shelving or shared rooms just don’t cut it.
Many people skip reading storage guidelines, storing compounds in sunlight or letting bottles crowd shelves. Heat and light stir up these chemicals, sometimes making them harder to handle or destabilizing them. Keeping original labels, tight lids, and cool, dark spaces sounds basic—until a spill or sudden reaction hits someone’s hands. Mistaking this caution for paranoia means missing the real risk: chemicals don’t forgive shortcuts.
Spills seem minor until the wrong rag spreads fine dust across a surface or someone tries to sweep it up with a brush. The rule I learned: never dry-clean. Dampen towels, trap the powder, and bag them right away. Even a forgotten puddle can release vapors for hours, outlasting a short lunch break. Friction or static make powders float—one more reminder to ground yourself and never trust old habits with new hazards.
No checklist or Notion document replaces experience, and repeating safety briefings builds a habit that gets automatic. Open questions about changes to chemical routines keep everyone sharp, especially with risky organics. In my circle, mistakes came not from ignorance but from confidence drifting too far from curiosity. It pays, every time, to listen to uncertainty—from coworkers, safety data, or your own gut.
Walking into a pharmacy or a research lab, shelves often show labels that sound more like puzzles than names: 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone is a perfect example. For those of us who have spent time pouring over molecular models in college, the name itself cracks open the formula. This compound carries a chlorinated benzene ring connected to a pyrazolone core. Every part of this name speaks volumes about how atoms fit together and how they shape what this molecule can do.
The molecular formula for 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone comes in as C10H9ClN2O. A benzene ring holds a chlorine atom at its second carbon spot. This ring tethers to the first position on a pyrazolone ring. Pyrazolone itself is a five-membered ring with two nitrogen atoms, one of which shares a double bond to an oxygen atom. A methyl group sits out at the third position. Chemists like to sketch this shape not just because it looks neat, but because the position of every atom changes what the compound can tackle in the real world.
Many of us might have taken a painkiller and not even thought about the scaffolding behind the medicine. Compounds like 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone make a difference because of their arrangement. The chlorine group resists easy breakdown, helping the molecule stick around longer where it’s needed, but also requiring careful handling in the environment because such bonds resist nature's cleanup crews.
The methyl group brings added bulk and bends the molecule’s shape in a way that can change its biological activity. A slight nudge in where the atoms sit or which group gets attached turns a simple compound into something medically active—or into a candidate for further tinkering in drug design. In many cases, pyrazolone derivatives paved the way for antipyretic and analgesic agents, making everyday life more comfortable for millions. None of this happens by accident. Scientists lean in close to these structures, tinkering, testing and searching for effects that matter in the clinic or in industry.
While the rings and branches might look plain on a textbook page, what happens after the compound leaves the bottle remains a bigger question. Chlorinated aromatics rank among the molecules that persist in air, water, and soil. Countries have seen these compounds travel far from where they were used, winding up in places where we never intended them to go. Regulators and scientists share concerns about how we monitor, treat, and manage compounds that won’t vanish quickly.
My own time in environmental chemistry taught me that small upstream changes in a molecule’s design can head off headaches much later. Swapping in an atom during the synthesis step, reformulating based on lifecycle analysis, or chasing biodegradable analogs—these small fixes can lead to cleaner rivers and safer disposal. Open data on molecular persistence and breakdown routes provides not just transparency, but a map for smarter decisions in the lab and beyond.
Knowing the exact placement of chlorine, the shape of the rings, and the role of the methyl group guides responsible chemistry. For students, practitioners and decision-makers, digging into the details of 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone means finding ways to unlock its benefits, balancing them with long-term safety and environmental impact. The molecules tucked inside everyday products deserve scrutiny, and the tools to understand them grow sharper each year.
Any time chemicals find their way onto shelves—be it at a research bench, a teaching lab, or a small-scale specialty facility—safety turns from theory into daily habit. 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone takes a seat among those compounds where careful storage matters. It's not something you tuck beside your groceries or leave unattended on a desk. Over the years in laboratories, even small oversights—wrong bottle, unnoticed leaky cap, container set near sunlight—have been enough to teach that even one weak link can expose both people and property to unnecessary risks.
If a bottle claims a spot among other reagents, the first step is simple: keep it somewhere dry, cool, and away from light. Many organic compounds, especially those carrying halogen groups like chlorine, react poorly with moisture. Water in the air prompts breakdown or changes in chemical structure. Heat sometimes nudges molecules into releasing vapors or changing state, and light—especially the harsh glare common near laboratory windows—can spark unwanted reactions or slow decomposition.
My own experience reminds me that people often underestimate the influence of indirect sunlight. A bottle on the wrong shelf, even half a room away from a window, still can absorb enough UV rays to discolor labels and, sometimes, affect the compound inside. Darkness gives molecules peace of mind. A sturdy amber glass bottle, tightly capped, tends to keep a compound from interacting with the outside world. Good habit goes a long way here—if you ever see powder around the neck or lid, take it as a hint that something needs fixing.
Chemicals like 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone should always stay clear of acids, bases, and oxidizers. Keeping jars spaced apart and clearly labeled avoids mix-ups. In shared spaces, labels with full names (not abbreviations) make sure anyone picking up the bottle understands what they're handling. It doesn't take much—just one misplaced vial in a drawer for hours of work to unravel over a spilled or misidentified chemical.
Ventilation makes a difference, too. I've seen situations where a sealed container in a closed cabinet still seeped odor, hinting at slow vapor release. Even solids sometimes give off enough to bother the nose or trigger headaches if left unventilated. Storing containers in fume hoods or ventilated cabinets, especially for chemicals you haven't used before, narrows the chance for an unpleasant surprise.
Keeping chemicals secure shouldn’t depend solely on rigidly following rules, but on understanding why those rules exist. If students or colleagues know what happens when dry storage slips, or why each hazard label matters, they look out for each other. Documentation—date received, expiration date, last inventory check—helps keep tabs on every container. Old or unknown powder shouldn’t hide in forgotten corners, either. Regular inventories, disposal of out-of-date chemicals, and shared responsibility have kept more than a few disasters at bay.
Safe storage isn’t about paranoia, but preparation. It’s an ongoing habit built from small daily decisions. Clean shelving, sensible segregation of chemical types, and good housekeeping—in my mind, these become just as crucial as the gloves on our hands or the goggles over our eyes.
Many chemicals show up in labs and factories, and 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone sits among them. Some people know it better as a building block for other compounds, and those on the production floor or in research labs want to know what they’re dealing with. Looking past the complex name, the real problem comes down to safety—what happens if this chemical slips out of its bottle or lands where it doesn’t belong?
A big concern with 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone revolves around direct contact. Breathing in dust or fumes can irritate airways. Overexposure causes headaches, coughing, sore throat, and sometimes dizziness. If it lands on skin, especially for long periods, it may drive rashes or swelling. Splash in the eye brings stinging, watering, or blurred vision.
Some pyrazolones have triggered allergic reactions. The body remembers the first encounter, and exposure down the line starts swelling, redness, or hives. For people with asthma or respiratory illnesses, dust or vapors might push symptoms further. While published studies do not single out this compound as a cancer risk or reproductive toxin, research on other chlorinated organics suggests caution. Long-term or repeated breathing in chemical dust can increase risk of chronic lung problems. Until someone proves absolute safety, following the rule of “least exposure is best” brings a lot of peace of mind.
Lab spills or poor storage can send this chemical straight into soil or water. It doesn’t break down quickly, and some research points to toxic effects in aquatic life. Runoff may threaten local streams or ponds, with the substance lingering and accumulating in plants and small creatures. Fishermen and swimmers near release sites have a right to worry.
Open burning or poor waste treatment adds another layer of risk. Combustion often releases toxic gases or byproducts, including chlorinated fumes, which harm both workers and neighbors. Safe disposal is not a small detail; hazardous waste rules protect not just the immediate team, but communities down the road.
Working near or with 1-(2-Chlorophenyl)-3-methyl-5-pyrazolone means using gloves that don’t rip easily, sturdy goggles, and masks suited for chemical dust. Spills need fast action—containment, cleanup, and good ventilation go hand-in-hand. Safety showers and eyewash stations right nearby make all the difference if plans go wrong.
Shifting culture in labs and on factory floors matters, too. Regular training actually keeps accidents from turning into headlines. Reporting close calls and learning from them weaves safety into daily work. Thorough documentation and real-time air monitoring pull their weight in busy settings.
Authorities in many countries already keep an eye on chlorinated organics. Inspections, reporting requirements, and permits force companies to take waste and emissions seriously. Good management follows these rules to the letter, even when no one’s watching.
Facing a chemical hazard like this, no single gadget or law fixes everything overnight. The right mindset—vigilance, transparency, and respect for risk—protects both workers and the environment. That’s the best safety legacy the chemical industry can offer.