Curiosity has always fueled advances in chemistry. The story of 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone springs from the broader search for new molecules with pharmaceutical value and utility in organic synthesis. Researchers working with heterocyclic compounds, especially in the middle of the 20th century, started exploring the pyrazolone core because of its promise for anti-inflammatory and analgesic agents. Chlorine substitution, like at the 3’-position on a phenyl ring, didn’t just happen by accident. Scientists observed that tweaking aromatic rings could shift biological activity, stability, and other properties. The emergence of this specific compound happened at a time when drug discovery depended on hands-on experimentation, trial, and often a good deal of patience. My time spent in chemical archives and literature searches brought me face-to-face with the sheer persistence in early medicinal chemistry, especially before combinatorial chemistry and high-throughput screening made such work routine.
Ask any chemist who has wrestled with naming conventions, and they’ll tell you that 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone reflects the logic and headache of IUPAC nomenclature. This molecule shows up in databases under several aliases, including 3-Methyl-1-(3-chlorophenyl)-2-pyrazolin-5-one and similar variants. Often, specialists know the same substance by multiple names depending on the application area, adding a strange kind of folklore to industrial and academic chemistry. Its core, the pyrazolone scaffold, crops up in everything from dyes to medicinal agents—though this particular variant has drawn more attention in research circles than as a household-name chemical.
Most of my experience in chemical research comes down to the grind of determining solubility, melting point, and stability, and this compound fits that mold. It usually appears as a crystalline, pale solid, shedding light on its aromatic backbone and fairly strong internal bonding. The presence of a chlorine atom at the 3’ position doesn’t just add mass; it alters how the molecule interacts with solvents, increases its density, and gives it new reactivity routes, especially in nucleophilic substitution. The methyl group at the 3-position of the pyrazolone ring nudges both electronic and steric properties, potentially influencing binding in biological systems. In the lab, one can’t overlook the need to handle aromatic chlorides with an eye for possible toxicity or environmental impact. I’ve watched less experienced chemists neglect personal protective gear, usually learning the hard way after just a few experiments.
In professional environments, the main thing technical documentation hammers home is practical purity, batch consistency, and clear identification. Certification usually involves melting point determination, chromatographic profiling, and spectroscopic identification. Forget fancy talk about “enabling” industry trends—on the bench, purity means real results. In research projects I’ve supervised, poor labeling or unclear technical sheets caused confusion and waste, underscoring the absolute need for tight specification and ongoing training in chemical hygiene. Chemists have debated signal wording, but straightforward hazard labeling with clear risk statements remains a necessity, especially for compounds like this where inhalation or skin contact carry hazards.
The preparation of 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone doesn’t stray from classic synthetic organic chemistry routes. Most approaches begin with condensation of 3-chlorophenylhydrazine (an aromatic hydrazine derivative with demonstrated hazards) with acetoacetic ester or its equivalents under acidic or basic conditions—proving that many new molecules spring from a simple, elegant logic. My college days included supervising undergraduate syntheses; I noticed that even slight changes in pH, order of addition, or reaction time produced striking changes in yield and byproduct profile. No two runs looked exactly the same unless the chemists developed a knack for observation and adjustment. The resulting raw product often required recrystallization to reach acceptable purity, driving home how much labor stands behind each “simple” white crystal.
This compound isn’t just a static presence in the chemical catalog. Its pyrazolone core offers many doors for derivatization. The chlorine on the aromatic ring invites cross-coupling reactions—Suzuki, Heck, or Ullmann—for the synthesis of new, more complex molecules. The methyl and pyrazolone positions offer possible sites for further acylation, alkylation, or oxidation, depending what scientists want to achieve. These types of transformations serve real-world needs—building pharmaceuticals, dyes, or specialty reagents. I’ve seen failure and progress hand-in-hand in attempts to coax new properties from subtle changes, emphasizing that chemical exploration has always been more about stubbornness and trial than magic bullets.
Chemists love to invent, and nowhere does this show up clearer than in naming conventions. Over the course of a career, I’ve seen the same compound go by half a dozen names, with old-school veterans favoring legacy terms while digital databases enforce rigid structures. For 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, one might run into synonyms like 3-Methyl-1-(3-chlorophenyl)-2-pyrazolin-5-one, or misspellings that crop up in legacy literature. This patchwork naming has often delayed literature searches and contributed to research inefficiencies. Databases and digital libraries still have work ahead to make synonym mapping more user-friendly and less error-prone.
Laboratory safety never gets old. Experience tells me that strict adherence to safe handling protocols isn’t just bureaucratic red tape. The presence of aromatic halides, potential for skin and eye irritation, and inhalation risks require not just gloves and goggles but proper ventilation and scraper discipline during clean-up. Mishandling, accidental spills, and improper waste disposal rank among the most preventable laboratory injuries. Training and regular safety reviews save more trouble than they cost in time. Regulatory requirements, from the local fire code up through international chemical shipping standards, all circle back to protecting the human element in chemical work—something that can slip through the cracks under deadline pressure.
Most discussion about 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone centers on its presence in research settings, primarily as a building block in synthetic organic chemistry and potential pharmaceutical intermediates. Scientists chase molecules like this for two reasons: seeking new drug candidates for conditions ranging from inflammation to neurological disorders, and building complex organic architectures to test hypotheses about biological activity. Sometimes, derivatives of this base compound edge into dye chemistry, pigment development, or specialty reagents for analytical chemistry. My collaborations with process chemists and medicinal teams always circled back to the time and resource hurdles just to move a scaffold like this from “interesting” to “useful” in any mass-market sense.
Chemistry never stands still. The trajectory of 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone research mirrors broader moves toward targeted drug design, computational modeling, and greener synthetic pathways. Machine learning aids in predicting novel derivatives with improved activity or lower toxicity, but in practice, real-world synthesis and biological testing still set the pace of progress. Academic labs push boundaries on functionalization schemes, using this compound’s core as a template for more potent or selective agents. In pharmaceutical R&D, improvements in yield and safety, reduction in hazardous byproducts, and the development of scalable synthesis methods play larger and larger roles in bringing ideas from bench to bedside.
Nobody wants surprises in toxicity research. Aromatic chlorinated compounds have, over the years, proved to be a double-edged sword—offering useful biological activity with real potential for harm. Animal studies and in vitro assays test everything from acute toxicity to chronic exposure, mutagenicity, and breakdown pathways. Most seasoned chemists, myself included, advocate for a cautious approach: treat unknowns with respect until the data arrive. Modern regulatory requirements press for full disclosure of adverse findings, making it harder to sweep side effects under the rug.
The biggest advances ahead for this compound lie in streamlining synthesis, tuning safety, and linking deeper biological knowledge to structure changes. Chemists and engineers seek to swap hazardous reagents for benign alternatives and reduce the brute-force energy inputs of yesteryear. At the same time, pharmaceutical research keeps one eye open for breakthrough activity among thousands of analogs that get made and tested each year. In my view, ongoing investment in predictive modeling, analytical improvements, and collaboration between academic and industrial research will determine whether this molecule becomes a stepping stone to new therapies, better materials, or just another twinkle of possibility in the chemical universe. The story here speaks to the patient progress of science—and the sheer tenacity it demands from those who walk its winding path.
Most folks have never heard of 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, but its name pops up in labs, textbooks, and industrial settings more than you’d think. In the simplest terms, chemists lean on this compound because it holds a unique spot in the field of organic synthesis and dye manufacturing. For those outside the science community, its use connects people to things they see and use every day—think colorants, inks, and various chemical products.
This chemical has found its place in the creation of vibrant pigments, especially ones used in specialty inks and textiles. Companies often use it to craft azo dyes, which land in everything from textile printing to plastics coloring. Growing up around textile mills, I saw firsthand how color mattered to both quality and sales on the final shelf. Getting colors right in dye batches isn’t just eye-catching; it’s a financial game changer.
Public health debates sometimes swirl around coloring agents in consumer items, but regulations on pigment safety have tightened over the years. The use of building blocks like 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone underlines the need for transparency in chemical sourcing and safer processing techniques. The textile industry keeps moving towards lower-impact dyes, but older compounds such as this one don’t leave the scene easily because they keep delivering what is asked of them: reliable, strong color.
Researchers also rely on this molecule for its reactivity. Organic chemists use it as an intermediate, which means it’s blended or modified to help stitch together more complex drugs, pigments, and advanced materials. The flexibility of the structure means you’ll find it in academic papers and patents on everything from drug development to analytical chemistry.
Years ago, my own science coursework ran into compounds like this during advanced synthesis, and I saw the careful steps needed to control reactions and cut down waste. People in chemical labs push for processes that limit exposure and waste because tighter rules and safer methods pay off over time. Green chemistry and better ventilation both matter more today, and experts agree: reducing the risk tied to chemical processes makes sense for workers, neighborhoods, and the industry as a whole.
The continued use of legacy chemicals like 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone points to a wider issue: slow progress in replacing older compounds. Many chemical businesses run on tight profit margins, and safer substitutes sometimes come with new headaches—like higher costs or altered performance. Industry leaders need to invest in research for alternatives and educate workers on proper handling.
To stay responsible, companies should keep tracking the environmental impacts and potential health risks of these compounds. Pulling in more rigorous third-party testing, and making the findings public, could build trust. Pressing down on chemical emissions and encouraging customers to ask about what’s really in their dyed goods nudges the industry in the right direction. Every step toward cleaner chemistry matters, both for the people making the stuff and the planet that absorbs it all in the end.
Anyone who's handled chemicals, whether in school labs or research facilities, knows an overlooked bottle can turn a busy day into a downright dangerous one. Storing compounds like 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone takes more than a half-closed cap and a shelf in the back room. The rules exist for a reason, and science, not just policy, backs them up.
This compound falls into a category where temperature, light, air, and moisture really make a difference. Over time, I’ve watched colleagues struggle with damaged reagents because the basics got skipped. Chemicals stored near windows or in humid spaces change character fast. 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone doesn’t love sunlight—direct rays push more energy into chemical bonds, sometimes leading to breakdown or new, unexpected products. Fluctuating temperatures only ramp up this risk, which is why researchers stick to cool, stable environments. Refrigerators with clear labeling become key tools, not just kitchen leftovers.
Humidity sneaks up on storage solutions too. Even if a container feels sealed, a cheap cap can let air and moisture work their way in, shifting purity or even altering safety. Anyone who’s opened a clumped or yellowed bottle knows the disappointment of water getting where it shouldn’t—especially when budgets already stretch thin. Dry, airtight containers paired with desiccators or silica packs help keep a pyrazolone derivative dry, and that’s not negotiable.
Chemical storage is a team exercise. People at every level, from undergraduates to postgraduate researchers, share the responsibility. Labeling with the name, date, and hazards avoids confusion. Clear hazard symbols serve a purpose—one glance should sum up whether the molecule inside reacts to air or light, or if it needs to stay away from acids or bases. It’s a simple habit, yet it stops mistakes before they snowball.
Real trust in any research group gets built on transparency about inventory. Someone finds a leaky seal or a cloudy solution, flags it, and the group takes action. Good practice follows federal and local regulations for chemical storage. Agencies like OSHA spell out storage for hazardous substances, drawing on lessons learned from real accidents. In my experience, labs that build safety into the routine—checking logs, training each newcomer, rotating chemicals to avoid expiration—tend to keep their doors open and their reputations strong.
Good storage keeps projects running. Every lost gram from poor handling cuts into experiments that cost both time and money. Anyone who drafts a grant proposal or teaches new scientists knows that chemical waste feels like watching opportunity go up in smoke. Smart buying, using only what’s needed, and storing the rest right should be second nature. Communicating across teams about needs and leftovers means everyone can focus on results, not damage control.
Driving improvements doesn’t hinge on expensive tech or fancy alarms; it’s about fostering careful habits. Investing in tight-sealing vessels and proper locations, teaching why conditions matter, and running regular checks make a world of difference. Everyone benefits when every substance—pyrazolone-based or otherwise—ends up in the right place, ready to deliver results, not surprises.
The world doesn’t usually pay much attention to odd-sounding compounds—until there’s a question about what they might do to our health. 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone might not sound familiar to folks outside a chemistry lab, but compounds like this show up across many industries. With its tongue-tying name, it’s easy to forget that real people work with and live around chemicals like this, and the risks are often overlooked until someone gets hurt.
A lot of these pyrazolone-derivative compounds end up in research, specialty manufacturing, and sometimes even in products that eventually reach the public. Chemicals with a chlorinated ring, like this one, can cause problems if handled carelessly. Contamination risk grows when companies cut corners or people lack the right information. Personally, I remember factories in my hometown handling chemical barrels—labels worn unreadable, with not a glove in sight. Even though this happened years ago, that worry sticks, mainly because safety slips are never just someone else’s problem.
So what makes a compound like 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone hazardous? Start with the basics: any molecule including a ‘chlorophenyl’ group can become toxic if it interacts with the body in the wrong way. Chlorinated aromatic rings have a reputation for sticking around in the environment, piling up in soil and even making their way into groundwater supplies. The methyl-pyrazolone segment adds another layer of chemical activity, raising concern about how this stuff breaks down inside the body or outdoors. Toxicity often comes from the body’s attempt to metabolize a substance into something harmless, but sometimes that process creates even nastier byproducts.
Open any industrial safety manual or toxicology review, and warnings pop up about compounds with similar structures. Repeated exposure can irritate the skin; some related pyrazolone substances are flagged for even more serious effects including liver problems or blood disorders. Agencies like NIOSH and OSHA urge caution with similar chemicals, emphasizing long-term effects that do not always follow an obvious pattern—chronic illness, slow poisoning, and subtle neurological changes.
None of this is just theory. In cities where chemical facilities operate without transparency, neighbors sometimes see the fallout firsthand—strange smells, rashes, unexplained nausea. Undetected leaks and shoddy disposal practices have left a mark in places that once thrived on industry, but later faced a public health crisis nobody saw coming. Trust evaporates fast in communities scarred by environmental hazards, especially if health officials and companies lag behind on reporting real risks.
It doesn’t have to go that way. Real safety starts with making sure everyone—researchers, plant workers, local authorities—has full access to every bit of available data about chemical hazards. Regulations matter, not as hoops to jump through but as basic protections for everyone downstream, literally and figuratively. Strong rules on labeling, stricter storage, better worker training, and investment in non-toxic alternatives can go a long way toward reducing harm.
People deserve to know what they’re being exposed to, whether in their workplace or downwind in their neighborhood. Industry can innovate without putting people at risk, but only if companies take responsibility for the chemicals they use. Clear, honest communication and tough enforcement work better than wishful thinking when it comes to issues of public health and safety.
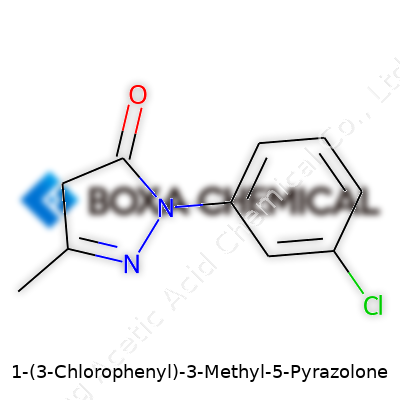
In labs and classrooms, I’ve watched how a chemical structure can change how people see a compound. With 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, the name already hints that something interesting is going on at the molecular level. You get a backbone of pyrazolone, which belongs to a family known for interesting pharmaceutical potential. The “3'-Chlorophenyl” part means there’s a phenyl ring—basically a benzene—tagged with a chlorine atom one spot over from where it meets the rest of the molecule. Add a methyl group at the third position, and you’ve got a shape that’s not just unique but meaningful in practice.
Let’s build the structure in simple terms: start with a five-membered pyrazolone ring, which carries two nitrogen atoms at non-adjacent corners. Connect that to a phenyl ring, then drop a chlorine at the meta (3’) position. Throw a methyl at position 3 on the ring. For chemists, seeing that chlorinated benzene ring brings to mind both added reactivity and the possible push-pull interactions that drive lots of changes in how a molecule acts. It’s more than pretty diagrams in a textbook—it’s about how those atoms talk to each other.
Take it from someone who’s spent hours breaking down molecular formulas: molecular weight isn’t just a number. For 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, it stands at 234.67 g/mol. This value guides everything from the amount needed to prepare a reaction, to how a molecule interacts inside a living organism. A change in molecular weight by just a few grams per mole can mean a totally different behavior in the body, which every medicinal chemist will tell you is not trivial. Dosing, solubility, and metabolism all hinge on that number.
In my own lab years, I watched colleagues agonize over calculations—checking and double-checking that one decimal point that could throw off a batch. Setting up a synthesis or figuring out toxicology demands absolute certainty about molecular weight because the math makes or breaks the experiment.
The structure and weight support lots of development in drug discovery. Tweaks to shape or the atomic lineup have led to painkillers, anti-inflammatories, and more. Students sometimes shrug off the importance, but real breakthroughs come when you respect just how much power lives in a tiny ring or side group.
The pharmaceutical field, in particular, has plenty of hard lessons about why a single atom can be the difference between therapy and danger. History recalls several drugs where a swapped atom meant new side effects or a lack of potency. For 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, the chlorine substituent and the methyl at position three probably emerged from a long process of evaluating what works and what doesn’t—often involving years of testing and observation.
Better understanding of molecular structure points to designing smarter drugs with fewer side effects. Chemists have tools for looking at how functional groups on a molecule might interact with enzymes or receptors. I’ve learned that cooperation between disciplines—synthetic chemists, computational folks, and clinicians—shapes safer and more effective compounds. The challenges include balancing the desirable effects with risks, especially as sometimes a structural tweak unlocks new, unintended activities.
If students and researchers focused more on those pieces—structure and weight instead of memorizing names or categories—they’d be set up for more creative, breakthrough science. The story behind 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone stands as a teachable example. At the end of the day, the combination of atoms shapes not only the molecule but every application downstream, from synthesis benches to clinical trials.
Anyone who’s had to wrestle with organic compounds in a lab knows that understanding solubility is not just for chemists with fancy titles. It’s a question that can decide whether a project stalls or moves ahead. 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone is a mouthful by name, but in practice, anyone looking to use this compound faces a practical question: Which solvent actually works?
From years spent in labs, I've seen that aromatic and heterocyclic compounds usually dodge water, asking instead for something that can handle a bit of nonpolarity or moderate polarity. It’s not surprising, then, that 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone often refuses to budge in plain water.
Instead, solvents like ethanol, methanol, dimethyl sulfoxide (DMSO), and acetone pull their weight here. Ethanol, for instance, brings out a good combination of safety and solvent strength. In research labs, DMSO often gets relied on — though its unique smell lingers everywhere — because it can handle both polar and some nonpolar compounds. And acetone? It evaporates fast, cleans glassware, and dissolves a surprising variety of chemicals, this one included.
Choosing the solvent isn't just bean-counting in a lab setting: it shapes safety, environmental impact, and cost. Solvents like DMSO unlock applications but stick to everything, so minimizing exposure keeps headaches (literally and legally) away. Ethanol offers a friendlier safety profile but comes with flashpoint limitations. Even small differences show up in waste bill totals, flammability concerns, and how complicated disposal gets.
Some solvents, such as chloroform or dichloromethane, also dissolve 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone, but few lab techs like wrangling those unless absolutely necessary. Personal experience has taught me that chasing easy solubility isn’t worth the trade-off: nasty fumes and health risks put a damper on productivity.
Textbooks and chemical supply houses list polar aprotic solvents and moderate alcohols as the best bets here, and published studies studying pyrazolone derivatives bear this out. Choosing solvents with an eye on practical workflow — not just theoretical compatibility — pays off. For instance, the fact that methanol and ethanol not only dissolve the compound but clean up without much fuss adds a dose of convenience.
Innovation keeps driving chemistry forward, and that trend turns up even when hunting for better solvents. Green chemistry aims to ditch high-toxicity options for safer, biodegradable alternatives; for researchers, industry techs, and students alike, pressure grows to pick less hazardous compounds even if it means tweaking old protocols.
Solubility doesn’t only shape experimental yield — it calls to mind ethics, resource management, and responsibility to others. Choosing solvents like ethanol, methanol, or DMSO for 1-(3'-Chlorophenyl)-3-methyl-5-pyrazolone often balances lab efficiency with greater good: protecting people and the environment. It’s worth remembering every time the next experiment gets set up.