Chemistry often finds its breakthrough moments during times of real need, and 1-Phenyl-3-methyl-5-pyrazolone traces its origin to a period when chemists sought better analytical tools. Discovered in the late 19th century, this compound answered the call for reagents capable of assisting in metal analysis—a crucial job as industries grew more complex. The name might sound esoteric, but for anyone working in analytical labs, it carries weight. By enabling rapid, reliable detection of metals such as iron and copper, it opened doors in industries ranging from mining to pharmaceuticals. Over the decades, researchers have shaped its methods of synthesis, purification, and use, reflecting a blend of scientific curiosity and practical demand. This story reflects the wider truth that solid progress in science often hinges not on flash but on steady, persistent tinkering and application.
In my own lab experience, 1-Phenyl-3-methyl-5-pyrazolone quickly showed its worth as a reagent that does more than sit pretty on a shelf. People outside chemistry circles might not recognize it, yet it’s a backbone in many tests and analyses. The molecule stands out in the crowded field of chelating agents. What grabs my attention is the ease with which it forms colored complexes, making metal detection much more straightforward. The fine, crystalline powder mixes into solutions with little fuss, and its strong performance in complexometric titrations put it ahead for chemists focused on precision work. Thanks to its versatility, this compound became a standard reagent in many analytical protocols, including colorimetric assays valued by environmental and food safety labs.
Handling 1-Phenyl-3-methyl-5-pyrazolone, I notice the white to yellowish hue and distinct crystalline texture—hard to mistake in pure form. Solubility leans heavily toward organic solvents, especially alcohols, which means keeping water out when prepping solutions is crucial. Unlike some reagents prone to rapid degradation, this one holds up with decent thermal stability, enduring basic storage conditions in cool, dry spots. Its molecular structure, with the aromatic phenyl group glued to a pyrazolone backbone, not only gives it stability but also fuels its ability to grab onto metals and form colored chelates. The melting point sits high enough to manage regular lab practice without special measures. For those who spend long hours with reagents, those small stability perks translate to a lot less hassle and waste.
Manufacturers supply 1-Phenyl-3-methyl-5-pyrazolone with clear purity ratings, usually notching up to 98% or above for analytical uses. Labels list batch number, purity, and lot analysis, which makes traceability easy—a sigh of relief for anyone dealing with regulatory audits or quality complaints. Some users overlook these details until a batch gives odd results or test failures start stacking up. Clear labeling might not excite the average person, but after a couple of mixed-up reagent disasters, I check labels compulsively. Proper packaging in airtight, light-proof containers keeps degradation and contamination at bay. The labeling often reminds users about dangers of ingestion or inhalation, and for good cause—responsible handling always begins with knowing what’s inside every bottle.
Formulation usually starts with phenylhydrazine mixing with ethyl acetoacetate, triggering condensation and cyclization steps common in classic organic chemistry. This core reaction does not involve complicated machinery or rare reagents, but timing, temperature, and pH call for real skill. Over many runs, I’ve seen how careful control turns a decent yield into an excellent one, and how sloppy preparation can leave behind unwanted byproducts. Crystallization from ethanol usually gives the purest product, but fast cooling sometimes traps impurities inside the crystal lattice. Experience counts for a lot here; what looks simple in a method booklet might need years of trial and error to nail down in practice.
The real beauty of 1-Phenyl-3-methyl-5-pyrazolone lies in its flexibility, both as a reagent and as a chemical platform. Lab chemists modify the pyrazolone ring to explore new metal-binding sites or boost selectivity for rare elements. Its chelating ability arises from both nitrogen and oxygen atoms in its structure, letting the molecule hold metals in stable complexes that change color in solution. In my hands, this color shift gives a sense of immediate feedback, invaluable for troubleshooting or rapid screening. Some researchers push boundaries by tailoring derivatives that fine-tune reactivity, opening up pathways in catalysis or synthetic organic chemistry. What emerges is not a static compound but a springboard for innovation.
Over years in academia and industry, I encountered this compound under different labels—phenyl methyl pyrazolone, PMP, and occasionally by its older trade names. These synonyms seem trivial, but confusion over names led to plenty of mishaps. Most scientific papers keep it formal, but distributors sometimes stick to short-hand versions. For those working across international teams, sticking to the CAS number makes communication easiest and avoids mix-ups especially in procurement or regulatory review. I learned never to assume a single name covers all variants; a quick double-check saves major headaches in the long run.
Lab safety around 1-Phenyl-3-methyl-5-pyrazolone always comes down to a blend of standard protocol and basic mindfulness. Inhalation, skin exposure, and ingestion all present real risks. Following guidelines, I suit up with gloves and safety goggles, and make sure all transfers happen under proper ventilation. Spills get cleaned quickly, since small amounts of dust can be irritating and persistent if left alone. Waste containers for pyrazolone residues need labeling for flammable and potentially hazardous waste—monthly audits at my old lab drilled this habit into everyone. Storage in sealed, original containers cuts down both risk and degradation, a move that saved more than one expensive batch from going bad before its time.
Few compounds I worked with cross the boundaries that 1-Phenyl-3-methyl-5-pyrazolone does. Analytical chemistry feels its strongest pull: water analysis, food safety, textile testing, forensic examination—all rely on its knack for signaling trace metals and contaminants. Hospitals once used related compounds in pharmaceuticals (like antipyrine derivatives for pain relief), but stricter safety data redirected these uses later on. In wastewater plants or mining labs, technicians count on it for colorimetric assays that catch pollutants before they harm ecosystems. The field keeps evolving, with more demand for sensitive, selective, and rapid tests. This compound, with its proven track record, often gets picked as the workhorse behind those solutions.
Current R&D focuses on expanding the potential of this pyrazolone through smart molecular tweaks. University teams keep synthesizing derivatives to improve specificity for rare earths or heavy metals, aiming for greener, faster analysis. Large-scale labs invest in automation that uses liquid-handling robots to streamline metal detection using this compound, reducing error and manual work. Few efforts strike me as more meaningful than the push for more selective chelators, especially as industries demand ultratrace detection in environmental samples. In applied research, interdisciplinary teams blend chemistry with data science, writing algorithms to read subtle color changes and translate them into numeric results—greatly reducing human error. Such integration marks a major leap for practical adoption outside specialized chemistry labs.
Published studies report moderate toxicity, especially via ingestion or sustained skin contact. Direct experience showed me how a small lapse in safety measures—like skipping a fume hood—could quickly lead to headaches or mild irritation. Some derivatives show higher risks, which keeps toxicologists busy mapping out the metabolic fate and breakdown products. Regulatory bodies flag repeated exposure for its potential liver and kidney impact, so chronic low-level contact is best avoided. Training new staff on toxicology details becomes a top priority. Animal studies help set exposure limits, yet a gap remains between lab data and real-world risks, especially for environmental persistence. That gap highlights the need for ongoing surveillance and transparent reporting.
Looking toward coming years, 1-Phenyl-3-methyl-5-pyrazolone stands poised for both expansion and competition. Fields like advanced sensor technology and portable test kits could see renewed adoption, especially as demand grows for low-cost, reliable chemical sensors in field work. On the other front, concerns about toxicity and environmental fate prompt calls for “greener” pyrazolone analogs, using bio-based starting materials or biodegradable substitutes. Synthetic chemists now partner with engineers to improve yield, purity, and safety, positioning this compound for a fresh wave of use in both established and emerging markets. My hope is that ongoing research closes the safety gaps while continuing to deliver reliable performance—so the blend of history, science, and responsible practice shapes the legacy of this venerable but adaptable molecule.
1-Phenyl-3-methyl-5-pyrazolone, widely called antipyrine, isn’t a household name, but its mark on medicine runs deep. Chemists first blended it up in the late 1800s, and it quickly made its way into pharmacies as a go-to pain reliever and fever reducer. Antipyrine has a knack for easing aches, so doctors favored it before newer painkillers came along. Hearing stories from older relatives, many recall their parents using medicines with antipyrine as an active ingredient to soothe ear infections or toothaches. The comfort people felt was real — direct proof that chemistry can bridge to well-being.
Most folks are familiar with aspirin or acetaminophen for everyday pain, but antipyrine still plays a role. Doctors sometimes reach for medications that mix antipyrine and benzocaine to tackle middle ear discomfort, especially after ear tube surgeries. Pharmacists trust the compound’s history of safety in these settings, leaning on decades of patient experience and clinical literature. It tackles inflammation at the source and calms nerves, so kids and adults alike get a little relief. That kind of impact holds value when you’re staring down a screaming toddler at midnight.
It’s not only humans who reap the benefits. Veterinary practices may use products with antipyrine for animals dealing with ear inflammation. The approach echoes what’s worked for people, proof that biology has a common thread, whether it’s a yowling cat or a child with an earache.
Beyond medicine cabinets, antipyrine carves out a niche in laboratories worldwide. Researchers and drug evaluators use it as a marker to check how the body processes and clears medication — a measure known as liver enzyme activity. Clinical studies track how fast antipyrine disappears from the bloodstream, helping professionals understand drug interactions and identify slow or fast metabolizers. Rather than relying just on new innovations, scientists see value in tried-and-true compounds that stand the test of reproducibility and reliability.
Old remedies sometimes fall out of favor not because they stop working, but because they get replaced by something that works better, or presents fewer side effects. Antipyrine doesn’t get prescribed as often as modern nonsteroidal anti-inflammatory drugs, but it hasn’t completely vanished for a reason: it still works well for certain patients and clinical needs. Regulators in many countries still allow its use, though doctors lean on clinical judgment and up-to-date guidelines.
Safety remains a concern whenever painkillers are in play, particularly thanks to adverse reactions or allergies. Anyone with a track record of sensitivity to pyrazolones should steer clear. Education among clinicians and open communication with patients can keep risks in check, though it’s up to health professionals and regulatory bodies to keep up with current research and flag new safety findings.
The story of antipyrine shows how science and history connect. Old drugs stick around for a reason, holding a lesson for how we treat pain and measure health. Newer isn’t always better in medicine; sometimes, the trusted standbys have an edge, especially when the evidence backs their safety and ability to help people feel better.
Few chemical names mystify chemistry newcomers the way 1-Phenyl-3-methyl-5-pyrazolone does. The name alone paints a picture of something complicated and almost intimidating. Put simply, this molecule holds real interest. The reason? It comes from its structure—one that links everyday chemistry lessons to discoveries in science, earning its stripes as more than just a pile of elements on a blackboard.
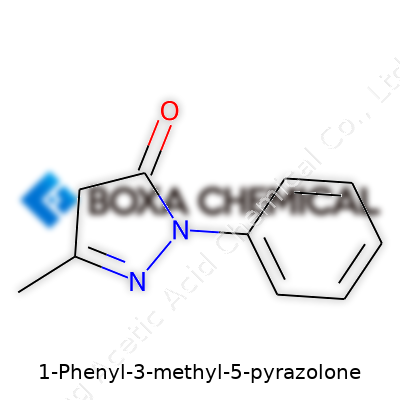
Draw it on paper, and 1-Phenyl-3-methyl-5-pyrazolone isn’t the most complex character in the lineup. Start with the heart of the molecule, the pyrazolone ring. This ring brings together five atoms—three carbon and two nitrogen—each sharing electrons to form a stable, flat arrangement that favors certain chemical reactions over others. Connect that ring to a benzene ring at position one, and snap a small methyl group at position three. These attachments don’t just serve as decorations. They change how the molecule behaves, how it binds with metals, and how scientists put it to work in the real world.
Structure shapes destiny. A molecule like 1-Phenyl-3-methyl-5-pyrazolone grabs chemists’ attention because its arrangement lets it act as a chelating agent. In my student days, teachers liked to talk about how this compound can grab metal ions—almost like a mitten grabs a hand. This feature turns it into a critical tool for both industrial analysis and academic research, helping measure traces of metals that matter in everything from water supplies to biological samples. The way its atoms fall together means it will form strong, predictable bonds—not by chance, but by design.
No one cares about a molecule just because it looks clever on the board. It’s the real-life stories that bring it to life. Those who work in environmental labs know that being able to reliably bind and detect metals determines public safety. The origins of this compound’s role in laboratories run deep, tracing back through hundreds of pages of scientific studies. Decades of research have uncovered how these atoms line up to make this molecule uniquely suited for analytics and even for creating special dyes and pharmaceuticals.
The old-school style of chemistry often focused on memorization, putting molecules into boxes and listing facts. With 1-Phenyl-3-methyl-5-pyrazolone, things get better with context. Understanding why the phenyl group at position one matters comes from watching how it changes solubility and reactivity. That methyl group at position three? It’s small, but it shields the molecule, making it easier to handle under tough lab conditions. These structural tweaks take this pyrazolone from just another compound to a workhorse of modern science.
Many problems arise in chemistry from a lack of real context. If students and researchers dove deeper into how structure drives usefulness, textbooks would move from rote learning into inspiration. Current challenges range from developing new sensors for environmental monitoring to building better drugs. Highlighting the design behind molecules like 1-Phenyl-3-methyl-5-pyrazolone can spark creativity. This comes back to good teaching and hands-on curiosity—something the structure of this molecule delivers in spades.
Real learning happens with real connections. With each nitrogen and carbon bond, 1-Phenyl-3-methyl-5-pyrazolone keeps teaching us that behind the name, there’s a story of usefulness shaped by the smallest of attachments. Pay attention to those details and new solutions begin to show themselves, both in the lab and far beyond it.
1-Phenyl-3-methyl-5-pyrazolone doesn't have the public profile of things like bleach or acetone, yet it pops up a lot in labs and some industrial settings. I still remember dipping into chemistry as a student, gloved hands shaking slightly as the instructor rattled off long names. Nobody in my class memorized this one, but plenty of us touched it at some point. So the big question—does it pose a danger? Is it another case where casual handling backfires down the line?
Let's start with the facts. This compound, also called PMP, draws plenty of attention in chemical circles due to its role as a reagent in analysis and as an intermediate in pharmaceutical and dye manufacturing. It isn’t known for explosive risks or extreme volatility, which tricks people into thinking of it as “the safe one.” But is it really?
Peer-reviewed chemical safety studies and materials safety data flag the potential hazards of PMP. Handling powders in high school labs, I learned pretty quickly that dust isn’t your friend, even if the powder looks harmless. 1-Phenyl-3-methyl-5-pyrazolone can irritate the skin, eyes, and respiratory tract. Accidental splashes sting, and the irritation can last. That stinging sensation sticks with you far longer in memory than in the skin. Respiratory exposure is worse, as inhaled chemical particles can cause coughing or worse, depending on the quantity and vulnerability of the person exposed. There isn't currently strong evidence linking PMP to long-term or cancer-causing risks in humans, but the lack of negative data shouldn't create a sense of false confidence. Absence of proof isn't proof of absence, especially with newer or less commonly used compounds.
I’ve seen people skip gloves because “It’s just another chemical.” Some even work without proper ventilation. This can go wrong quickly, as fine powder finds ways to reach eyes or lungs. Sharp medical minds suggest using protective equipment with any chemical capable of provoking a reaction, no matter how mild. You can wash out a splash in the eye, but not the worry or regret if something more serious develops.
The importance of good habits around chemicals stands out in every experience I’ve had. Lab coats, gloves, and goggles keep risk at bay for people using PMP. Routine cleaning and neat storage habits come into play, too. You don’t want search-and-rescue missions just to find spilled powder hiding in the corners. Airflow matters—without a fume hood or at least open windows, the air gets heavy, and irritants hang around longer than welcome.
Training goes a long way. Brief refreshers on chemical safety help prevent mistakes during routine work. Many companies rely on internal audits, peer-to-peer reminders, and required briefings to limit risk and build a culture of safety. Safety Data Sheets deserve a read before and not after a problem crops up; they lay out symptoms, protective steps, and first aid. I’ve never regretted taking five minutes to check safety notes before opening a new chemical bottle.
Respect for chemicals, even the ones labeled “routine” or “not especially toxic,” sets the tone in any workspace. Every person managing chemicals—at a university bench or in a larger plant—can make a real difference by building careful routines. With 1-Phenyl-3-methyl-5-pyrazolone, the solution comes down not to fancy technology but to consistent use of basic protective measures, a questioning mindset, and a refusal to cut corners, even on quiet days.
Anyone who’s spent time around labs or chemical storage knows it's not just about following a script. How we store a compound like 1-Phenyl-3-methyl-5-pyrazolone, also known as antipyrine, impacts research, safety, and our bottom line. I’ve learned the hard way that missing even one detail, like a slightly open jar or a shelf too close to a heat vent, can ruin an experiment or trigger a safety scare.
This compound doesn’t throw a fuss in daylight or at a typical room temperature, which makes it more forgiving than some other reagents. Most manufacturers settle on bottle labeling that advises cool, dry, well-ventilated storage, and experience backs that up. Heat and moisture chew away at shelf life, and nobody likes surprise clumps in powder or degraded results on an assay.
I’ve opened a few forgotten bottles of the stuff tucked near radiators, and what you get isn’t the stable powder promised on the safety sheet. It cakes, develops off-odors, and sometimes even discolors. That’s more than just an eyesore—it brings into question every result you pull from that supply. Cabinets set away from direct sunlight, moisture sources, and heat vents save time and money in the long run.
1-Phenyl-3-methyl-5-pyrazolone isn’t a notorious hazard like some volatile organics, still, it deserves respect. If moisture gets in, the compound may partially hydrolyze, diminishing purity. Given its common use in pharmaceuticals and dye intermediates, any dip in quality could have ripple effects. A friend working in pharmaceuticals once told me a single contaminated batch early in a production run sometimes means weeks of backtracking and lost revenue.
Many worry about fire risk, though the compound doesn’t ignite as easily as some solvents. Still, dust can become a respiratory irritant if mishandled. A forgotten cracked lid delivers unpleasant surprises—sneeze-inducing powder into the air and potential for reactions with anything acidic or oxidizing nearby.
Storing this powder in tightly sealed containers—preferably glass or chemical-resistant plastic—remains the gold standard. The old advice about desiccators and silica packs really pays off if you live in a humid city or work in a lab where air conditioning drifts in and out. Placing containers in secure, labeled cabinets, preferably locked, puts extra assurance that only trained hands access it.
Periodic checks go a long way. Too often, labs stock up for the year, then ignore what’s gathering dust. Set a schedule. Measure the appearance, record dates when you open new bottles, and never leave portions in unlabeled containers. These small acts keep everyone from guessing what’s inside or fearing contamination later.
The most impressive lab spaces I’ve visited don’t always have endless budgets. Instead, their teams show a kind of discipline—cleaning shelves, updating logs, throwing out poorly stored chemicals. It isn’t about perfection, but about giving each batch of 1-Phenyl-3-methyl-5-pyrazolone the respect demanded for quality science and safety. Good storage reflects serious work, and it pays back every single time with results you can trust.
Anyone working with chemicals in a lab or industry knows that purity is more than just a number on a sheet. Looking at 1-Phenyl-3-methyl-5-pyrazolone — often used as a reagent or intermediate — the purity of the compound points straight at safety, performance, and even research results. Too many people outside the lab may think a small impurity doesn't change much. Anyone who has run a reaction or built a process from the ground up learns otherwise fast.
Purity standards for this compound usually run above 99%, and for a good reason. Trace contaminants easily throw a wrench in chemical reactions, sometimes leading to unexpected byproducts or outright failure. Tiny deviations in purity have delayed projects because someone missed something during quality checks. That frustration lingers when running time-sensitive experiments or trying to deliver on a customer promise.
In pharmaceuticals and labs, purity often means “HPLC ≥ 99%” or “GC ≥ 99%”, supporting precise reactions and reducing headaches later. Reagents with these numbers have been put through chromatography, confirming what’s in the bottle matches the label. This level reassures users, who can rely on consistent results batch after batch. From experience, trusted suppliers rarely cut corners here — reputation depends on predictable outcomes. Labs, drug makers, and researchers want to see certificates of analysis to keep track of compliance and benchmarks.
Even a few tenths of a percent variation creates room for error. Many analysts I’ve worked with keep a close eye on the water content and residual solvents too, especially when the compound will be used downstream. Just last year, a colleague found that a single batch with a higher moisture content ruined a sensitive reaction sequence, forcing them to start over from scratch.
Anyone serious about using 1-Phenyl-3-methyl-5-pyrazolone looks past the sticker price straight to test reports. Regulatory bodies and health agencies have steadily tightened traceability and reporting standards. It’s no longer enough to take the seller’s word. Responsible labs demand up-to-date documentation on every batch, sometimes turning away suppliers who can't provide verified purity specifics.
Purity affects chemical storage, shelf life, and downstream use. No chemist wants to discover impurities after a project is underway. I've witnessed projects stall because the documentation didn’t match expectations or the batch had hidden contaminants. Serious organizations run their own quality checks, sending raw samples for third-party validation before using them in critical research or product development.
Sourcing high-purity 1-Phenyl-3-methyl-5-pyrazolone takes time. Relying on suppliers who publish their testing procedures and results — especially those that use reputable third-party verification — reduces risk. Full transparency and clear communication about testing methods give peace of mind. Where possible, storing chemicals under recommended conditions and re-testing at intervals can catch deterioration before it affects the work.
The industry still faces slip-ups — mislabeling, contamination during transport, or limits in local testing equipment. Communication is key. Users should reach out, ask for updated reports, and clarify anything that looks even slightly inconsistent. In my own work, addressing concerns early and keeping a strict paper trail has often paid off, dodging costly setbacks and misunderstood failures.