Ask anyone who’s spent time in an industrial chemistry lab about 2,3,5,6-Tetrachloropyridine, and you’ll probably hear a quiet respect in their voice. This compound didn’t just pop up overnight. Its roots stretch back to post-war advances, when the world ramped up demand for specialty chemicals. Chemists looking for more effective intermediates began to see pyridine rings, especially the chlorinated ones, as powerful building blocks. People found that swapping hydrogen atoms for chlorine on the pyridine ring—especially at the 2, 3, 5, and 6 positions—opened up a playground for industrial synthesis. This came during an era obsessed with better herbicides, pharmaceuticals, and even dyes. A glance at the literature from the 1950s onwards shows a steady trickle of research, reflecting a compound whose utility has kept pace with evolving industry demands.
With a sharp, almost acrid odor and a crystalline solid appearance, 2,3,5,6-Tetrachloropyridine stands out from other pyridines. Each chlorine atom changes the electron density of the pyridine ring, making it less reactive than the parent compound but far more useful as a starting point for synthesis. It’s not particularly volatile, and its water solubility compares poorly to simpler pyridines. These small changes in structure carry big weight in manufacturing, especially when companies want something stable but primed for further chemical changes. The unique substitution pattern on the ring gives it a balance of reactivity: resistant to full-on degradation, but open to specific modifications that broaden what chemists can create. Unlike some lab curiosities, this compound has real-world muscle.
2,3,5,6-Tetrachloropyridine’s molecular formula (C5Cl4N) and its tightly packed arrangement of chlorine atoms push its melting point well above room temperature, demanding robust handling practices in industrial settings. Its density and low volatility make spills less likely to vaporize, which may help limit immediate inhalation risk but shifts concern toward containment and disposal. The molecule’s chlorinated nature means it’s neither a shrinking violet nor a pushover—handling requires respect, adherence to good ventilation, and routine checks on personal safety equipment. Its chemical toughness means it rarely breaks down in the environment or inside exposed waste streams, which has drawn scrutiny from environmental chemists. I’ve seen labs give extra attention to waste collection for compounds like this, knowing all too well how persistent chlorinated aromatics can be if mishandled.
Making this compound often starts with pyridine, which undergoes sequential chlorination. More than one route exists, but most involve reacting pyridine with chlorine gas, using a catalyst and carefully controlling conditions so chlorines land in the right spots. It’s a delicate balance—too much heat or a slip in timing, and unwanted byproducts pile up. This is not a job for the careless. Tightly managed reaction vessels are the norm, often under strict temperature protocols to avoid accidental over-chlorination or even localized combustion. Each step must consider energy input, byproduct treatment, and purification methods—not just for efficiency, but also for the sake of downstream environmental concerns. Lab veterans know the process doesn’t just stop at the reaction; getting high-purity tetrachloro compounds cleanly out of solution often becomes an industrial-scale filtration and distillation puzzle.
2,3,5,6-Tetrachloropyridine isn’t a wallflower in synthetic organic chemistry circles. The electron-withdrawing chlorines dampen the aromatic ring’s basicity, yet leave specific positions open for substitution, especially under nucleophilic conditions. In practice, this means skilled chemists can introduce groups like amines or alkoxides, yielding highly functionalized derivatives that become stepping stones for crop protection agents or advanced dyes. Each modification opens doors to a different industrial need, from pharmaceuticals to agrochemicals. In my experience, the number of reactions and modifications possible with this compound make it an outstanding raw material for R&D labs probing new molecular scaffolds or making analogs of known products. It’s not a Swiss Army knife, but it performs its specialist role with a kind of efficiency that gets recognized in retrosynthetic plans across multiple fields.
You might also hear 2,3,5,6-Tetrachloropyridine called Tetrachloropyridine or by trade or catalog numbers in technical literature or chemical supply listings. Other researchers sometimes refer to it by shorter, systematic abbreviations in lab notes, but no matter the name, it’s the utility and reliability that keeps this compound in the toolbox. These alternate names tend to surface when discussing historical research or navigating older scientific databases. They point toward the dense network of applications and regulatory records that have followed this substance since its introduction.
Nobody with experience handling this material or similar chlorinated organics would call it harmless. Everything from lab benches to large-scale manufacturing setups comes with a checklist when this compound is present. Eyes and lungs don’t get a second chance, so chemical goggles, gloves, and fume hood work are non-negotiable. Because it can cause irritation and has flagged up some health concerns in toxicity reports, careful storage and clear labeling play a real part in cutting down on accidental exposure. Teams working with such materials usually invest heavily in safety drills, spill containment readiness, and strong air filtration. I’ve seen how adherence to local and international safety guidance not only keeps people working safely, but also reassures local communities. Good safety practice with this material isn’t a choice—it’s a responsibility built on decades of lessons learned.
Across industry, 2,3,5,6-Tetrachloropyridine pulls its weight, particularly as a key intermediate in pesticide synthesis. Large agricultural outfits know that active ingredients built from this molecule can change the economics of crop protection. Its presence shows up in other specialty chemical products and R&D paths, feeding into pharmaceuticals and even advanced materials research. The flexibility and reliability attach this compound to supply chains that value consistency and purity. As a regulator once put it to me, the ongoing attention to the environmental impact of such compounds hasn’t slowed down their use—only spurred a new wave of rethinking about how these ingredients are produced, deployed, and ultimately replaced.
Recent years have seen ramped-up research into potential health and environmental risks of chlorinated pyridines, including 2,3,5,6-Tetrachloropyridine. Concerns have arisen around persistence and potential for bioaccumulation, actions that often lead regulatory agencies to ask tough questions. Inhalation or skin exposure risks stand out most clearly in studies, with repeated emphasis on acute irritation and possible long-term effects. Ongoing toxicological profiles provide some relief—large-scale poisoning events haven’t shown up—but questions around trace exposures keep researchers busy. Environmental groups keep a steady pressure on manufacturers to develop safer handling and more robust containment systems. A few R&D teams have begun exploring green chemistry approaches, even reengineering routes to introduce less toxic starting materials and finding ways to close the loop on chlorinated byproducts.
Chemistry doesn’t freeze in place, especially not when safety, sustainability, and changing regulations push for smarter solutions. As new green chemistry routes mature, the hope is to replace some of the more hazardous steps in tetrachloropyridine’s production, making future supply chains cleaner and safer. Calls for transparency push the industry to publish cleaner toxicological data and lay out clearer labeling standards, helping both workers and downstream users make informed choices. The prospect of stricter environmental regulations lingers, nudging R&D teams to keep refining both the compound itself and the processes surrounding it. Industries that rely on tetrachloropyridine show no signs of abandoning it overnight, but every sign points to a quieter revolution—one that values environmental legacy just as highly as output or efficiency. This matters to everyone from chemists in the lab to the communities that live alongside chemical manufacturing hubs. Progress in handling, hazard reduction, and innovation may prove the biggest legacy this compound leaves behind.
Most people flip past the fine print on pesticide and chemical manufacturing labels, never thinking twice about the names listed. 2,3,5,6-Tetrachloropyridine rarely catches anyone’s eye, yet it keeps showing up deep in the supply chain for crop protection and certain specialty industrial goods. This chemical often acts as an intermediate, which means it’s never the final star of the show. Its real impact comes from the important stuff made from it—especially certain pesticides, herbicides, and specialty chemicals.
Sitting between the field and the food on our tables, 2,3,5,6-Tetrachloropyridine plays a supporting role. It can serve as a backbone for synthesizing popular crop protection agents, such as insecticides or herbicides. We depend on these substances to keep crops healthy, boost yield, and meet growing demand in supermarkets around the world. For context, modern agriculture rarely functions without chemical support. Practically every large-scale farm leans on treated seeds, pest control, and weed management. Most of the compounds doing that heavy lifting start as raw chemicals like this one.
I learned about these chemical building blocks back in a college organic chemistry lab. A yellowish liquid sat carefully labeled in a fume hood. My professor pointed out that compounds like 2,3,5,6-Tetrachloropyridine usually don't stick around until the final product. They break down or morph into something else long before any food leaves the factory. I never saw those complicated names in real life until covering agri-tech as a journalist. It turns out, companies use this stuff upstream to craft safer, more effective answers to natural pests and plant diseases. It rarely touches the soil in its original state.
Yet, there’s a catch. The process of making and moving these chemicals has risks. Mishandling them can lead to environmental and health hazards—both in the short term and long term. Workers on the chemical side face exposure dangers, and accidental spills threaten local ecosystems. Several years ago, local news reported wastewater contamination not far from an old synthesis plant. Cleanup taxes public budgets, and the headaches can last for years. This makes oversight critical, from manufacturing all the way to final product use in the field.
The world needs affordable, available food. Chemicals like 2,3,5,6-Tetrachloropyridine, even if mostly invisible to end consumers, form a link in that chain. Skipping them would mean relying more on old-fashioned, often less efficient methods. Yet, the tradeoff isn’t always obvious. Producing these intermediates puts strain on regulations. Good stewardship calls for clear labeling, improved waste management, worker protections, and investing in greener chemistry.
Some companies already look for ways to shrink the footprint left by intermediates like this. Closed-loop systems, advanced filtration, and real-time leak monitoring are nudging things forward. In academic circles, researchers are exploring biobased alternatives that tackle some of these synthesis steps without as many harmful byproducts. Progress tends to crawl in the chemical industry, but even small gains ripple outward.
At the end of the day, most people won’t ever remember the name 2,3,5,6-Tetrachloropyridine. Yet, what happens with chemicals like this decides a lot about what’s on our plates and what ends up in the environment. Staying alert to how these substances shape the future isn’t about doomsaying—it's just honest, common sense stewardship.
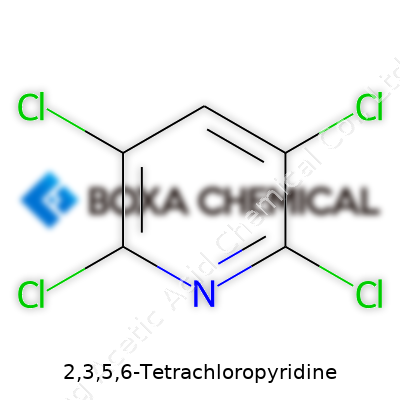
A chemical name like 2,3,5,6-Tetrachloropyridine doesn’t sound like something most people come across in daily life. But for chemists and anyone working with agricultural or industrial chemicals, this molecule matters. Its formula, C5Cl4N, tells a story. Five carbons, four chlorines, and a nitrogen—all tucked into a structure based on pyridine, but with four of its hydrogens swapped for chlorine atoms.
For anyone who’s spent time in a chemistry lab, formulas aren’t just codes. They give a window into what a chemical might do, what risks come with it, and how it can be useful or dangerous. For this one, the name tells you a lot. Take pyridine—simple, six-membered ring with a nitrogen. Add chlorines at the 2, 3, 5, and 6 positions. You get a compound that’s not just another bench chemical, but part of a larger toolkit for pesticide and pharmaceutical manufacturing.
Chlorine atoms bring a handful of changes to a molecule. They ramp up stability against breakdown, might boost or reduce toxicity depending on where they land, and can make a compound stick around a lot longer in soil or water. This isn’t just an academic observation. There’s a reason why many herbicides, insecticides, and specialty solvents use chlorinated rings.
Growing up around farming communities, I’ve seen firsthand how chemistry turns into crop yields—or contamination. Urban folks might think of the “chemicals in agriculture” headline, but rarely see the choices behind the spray. Chemicals like 2,3,5,6-Tetrachloropyridine bridge research and reality. When this molecule shows up, it’s usually because someone wants precision: kill a weed, stop a fungus, or build a more effective drug. The formula guides those choices.
C5Cl4N isn’t just a string of letters and numbers. It signals higher toxicity compared to its less chlorinated cousins. That raises questions about bioaccumulation, persistence, and safe disposal. Public health history has plenty of examples—take DDT or dioxins—where added chlorines made chemicals last longer and turn up in unexpected places. People living near heavy pesticide areas know the price of risky chemical handling.
Facts from research make a difference. Chlorinated pyridines show up in degradation studies, water sampling near industrial sites, and governmental chemical watchlists. Agencies like the EPA and EFSA pay close attention because a stable, persistent compound ups the stakes for wildlife and human exposure.
Society needs chemists and industry leaders to look beyond the formula sheet. C5Cl4N’s structure suggests both potential and risk. Safer handling demands proper training, strict limits on releases, and open data on where it turns up. Organic alternatives or less persistent molecules are possible for many uses, and sustainable chemistry pushes toward designing out legacy problems.
In the end, knowing the formula isn’t trivia. It means understanding the real-world impact of a single molecule—who it helps, who it might hurt, and how people can make decisions together for safer, better results.
Every so often, chemical names like 2,3,5,6-Tetrachloropyridine grip headlines with an air of mystery and warning. It crops up in industrial processes and research labs, yet its impact on health can feel foggy for people who don’t spend their days reading material safety data sheets. It’s fair to ask whether this chemical should worry anyone beyond lab workers or factory managers. Based on what’s known, it deserves a careful and honest look.
2,3,5,6-Tetrachloropyridine is a halogenated pyridine — that sounds like a mouthful, but in plain terms, it belongs to a family of chemicals often used as building blocks for pesticides, pharmaceuticals, and specialty chemicals. It doesn’t pop up on grocery shelves or household products, so regular folks rarely come across it in daily life. Most exposure risk lies with workers who handle the chemical during manufacturing, transport, or disposal.
From a safety standpoint, experience and science both show that chemicals packed with chlorine atoms often attract suspicion. Chlorinated organics, as a class, can be tough on the human body. Studies on similar substances show that prolonged or high-level exposure raises risks of harm — think, eye irritation, skin allergies, and damage to organs over time. Inhalation or accidental spills onto the skin can bring acute symptoms right away: headaches, respiratory discomfort, or rashes. The degree of danger depends on concentration, length of exposure, and whether the substance gets on skin, in eyes, or into the air as a vapor.
Long-term effects matter, too. Some chlorinated pyridines have sparked concerns over links to liver damage, kidney problems, and even cancer in animal studies. Records for 2,3,5,6-Tetrachloropyridine itself don’t show sweeping studies on people, so the scientific community relies on results from similar chemicals to set guidelines. Regulatory bodies urge high caution — groups like the EPA treat new or unexplored chlorinated compounds carefully, often classifying them as possible or probable hazards until proven otherwise.
Those of us who have spent time in labs or chemical plants know the importance of proper storage, ventilation, and personal protective gear. No one walks around “getting used” to working with solvents and strong reagents like this. Accidents even in well-run facilities have taught a simple rule: treat unknown or reactive chemicals as if they could harm you at low levels. Industry standards mandate safety goggles, gloves, lab coats, and chemical fume hoods for a reason. Skimping on these pays a steep price.
People deserve to work and live in environments where hazardous chemicals don’t stealthily creep into the air or water. Strong government oversight makes a difference. Agencies need transparent chemical registries and clear rules about emissions, disposal, and worker protections. Communities benefit from open access to information about nearby plants and the chemicals they use. Companies have a duty beyond profit: they ought to invest in training and equipment that keeps accidents rare.
On a broader scale, pushing for safer alternatives can change the game. As science advances, manufacturers should support green chemistry — picking substitutes that carry less risk to people and ecosystems. Shared experience through research and open reporting helps everyone make smarter choices, whether in industry or at home. Safety around chemicals like 2,3,5,6-Tetrachloropyridine rests on vigilance, education, and collective will — not just in reaction to headlines, but as a daily practice.
Anyone who’s spent time handling industrial chemicals knows the odd mix of respect and caution each new substance demands. 2,3,5,6-Tetrachloropyridine isn’t a household name, but in certain industries, it’s a necessary part of the toolbox. What matters most is not just knowing its properties, but understanding how to work with it without risking your health, your co-workers, or your neighborhood. This isn’t a mystery you solve with a few quick searches. Relying on science, industry reports, and practical experience shapes smart habits around it.
This is a chlorinated pyridine. That means it brings toxic and possibly persistent behavior to the table, much like other related compounds. Breathing its vapors or spilling it on skin has real consequences. There’s evidence it can cause irritation, and there are gaps in knowledge about all its long-term effects. A wise person never assumes a new chemical is harmless because it hasn’t made big headlines. Decades back, workers handling PCBs and certain solvent vapors thought similar things and paid a steep personal price. Staying in the safe zone takes more than routine glove use or half-hearted ventilation.
Storage means more than locking a drum in the back room. This chemical needs a well-ventilated space with no silly shortcuts—no storing it near acids, bases, or anything that could trigger a reaction. Temperature and moisture matter. Keeping it cool and dry helps limit both vapor release and breakdown. Imagine a spill or slow leak—it quickly turns from an issue for one worker to a challenge for everyone in the building and beyond. I’ve seen careless practices turn into insurance nightmares that stick around for years.
I always recommend using dedicated containment trays or cabinets. Label everything boldly. This ensures no one—visitor or regular hand—mixes things up. There’s a temptation to treat “known” chemicals with less care, but I’ve watched this blow up on more than one occasion. Responsible storage protects both people and businesses from slip-ups that lead to fines and worse.
Putting on gloves or masks isn’t enough if you don’t know exactly which kinds offer protection. Nitrile and neoprene gloves usually handle chlorinated organics better than latex. Eye protection stays on, splash shields go up before pouring. Anyone handling the chemical should get training—not just an online checklist, but a walkthrough from someone who’s actually dealt with incidents before. Real disasters rarely look like textbook scenarios; they unfold when distractions, assumptions, and incomplete prep come together. I’ve learned a lot from watching emergency drills turn up obvious gaps in supposedly airtight procedures.
Following rules on paper wins you no points if the team isn’t bought in. Clear, open communication about risks—without embarrassment or bravado—goes much further. Encourage reporting of near misses and reward careful workers, not just the fastest. Companies earn reputations not just by profit margins, but by how they handle hazardous materials. There’s pride in a shop with zero incidents year after year, and it’s usually no accident; it follows tight, common-sense management and relentless vigilance.
Government guidance on storage and exposure limits is there for a reason. Companies that ignore it sometimes find regulators knocking, but more often, the lesson comes too late in the form of preventable harm. Readers handling 2,3,5,6-Tetrachloropyridine or anything like it owe it to themselves, their teams, and their communities to treat every day like it’s the first day working with a volatile compound. Experience and humility make better neighbors than overconfidence ever could.
People with hands-on experience in labs or chemical plants know the worry that comes with a bottle labeled 2,3,5,6-tetrachloropyridine. That long name signals more than just tongue-twisting pronunciation—it’s a nudge to act cautiously. This chemical sits among those industrial substances with proven benefit to chemists and researchers, but it brings a level of risk not to be ignored.
Breathing its vapors or dust, or letting it touch the skin, steers you straight into trouble. Tetrachloropyridine contains multiple chlorine atoms, which tend to foster irritation and toxic effects. Prolonged contact can provoke skin burns or trigger nasty respiratory issues, and the compound’s volatility means even a small spill can fill a room with fumes. The toxic profile of halogenated pyridines, backed by published toxicology reviews, warns of dangers to both people and the environment. Workers exposed to similar compounds without adequate precautions have recorded symptoms from headaches to nausea to lung problems.
I learned my lesson a decade ago in a synthetic chemistry lab after seeing a careless colleague try to pipette tetrachlorinated organics without gloves. In less than five minutes, they experienced coughing fits and red eyes—a fast education for the whole team. It’s easy to be lulled by habits or by the illusion that “it’ll be quick and safe,” but chemicals like this refuse to forgive shortcuts.
Work always starts with personal protection. Lab coats, splash-proof goggles, chemical-resistant gloves, and fitted respirators create the first barrier. It’s practical, not paranoid. In my own work, I keep nitrile gloves on for all handling, and if I’m weighing or transferring tetrachloropyridine, I use a certified chemical fume hood. Open benches belong to another era; airborne hazards move fast and sometimes stay undetected until it’s too late.
A big safety step comes with knowing how to clean up. Spills come without warning. Absorbent pads and neutralizing powders for chlorinated compounds must stay close, and waste needs disposal in designated chemical containers—never the sink or regular trash. A friend in waste management hammered home that leaks hurt more than just the person holding the bottle—traces can contaminate water sources, so tracking and containing every drop matters. Federal and local guidelines on environmental protection serve as more than just paperwork—enforcement actions and fines spring from neglected spills.
Clear rules for labeling, storage, and restricted access keep these chemicals out of untrained hands. Small signs, locked cabinets, and inventory logs may irritate some workers, but they stop missteps before they happen. At the last firm I consulted, routine drills on chemical accidents gave people the muscle memory to respond instead of panic. Fire extinguishers suited for chemical fires and eyewash stations near benches lower the stakes if something goes wrong. Reliable ventilation systems save countless lungs from harm, and I always push for regular testing rather than trusting a fan installed years ago.
Most injuries I’ve seen come from shortcuts or inattention, not unpredictable accidents. It comes down to attitude: valuing safety as an investment, not as a hurdle. If each team member treats risk with respect, the odds tilt in everyone’s favor. Hazards tied to 2,3,5,6-tetrachloropyridine reflect a bigger story in chemical safety—real danger lurks behind the label, but practical habits supported by experience and facts build safer labs, factories, and communities.