Chemicals like 2,4-Dichloropyridine didn’t just fall from the sky. They have roots in the long journey of organic synthesis stretching back to the first reliable ways to introduce halogens onto aromatic rings. In the early half of the 20th century, industrial chemistry came alive with a hunger for new synthetic scaffolds for drugs and agrochemicals. Pyridines had already built a reputation thanks to nicotine and its relatives. Someone in the lab combined chlorine and pyridine, probably with short sleeves and no fume hood, and the family of dichloropyridines joined the chemical inventory. These compounds spread because researchers spotted their utility: the structure allowed chemical tinkering, and the chlorines gave the ring more muscle for downstream reactions. Decades later, 2,4-Dichloropyridine sticks around, embedded in chemical supply houses and research labs worldwide.
2,4-Dichloropyridine sits in that odd middle ground—a specialty chemical that’s never famous but quietly supports bigger categories like pharmaceuticals, crop protection agents, and dyes. This colorless to pale yellow liquid or crystalline solid has shown up in my own bench chemistry work as a go-to intermediate. Its pyridine core stands out because of those chlorine atoms at the 2 and 4 positions on the ring, and they make the molecule a prime platform for quick modifications. Labs order it by the kilogram for synthesis work, and though you won’t find it on grocery shelves, it’s behind the scenes in industrial products that help feed, heal, and clothe people.
I keep a mental image of 2,4-Dichloropyridine from those days in grad school: a slightly acrid odor, a density higher than water, and the reliable weight of a bottle that you know must stay tightly sealed. Its melting point and boiling point are mundane features on technical sheets but crucial in practice. The presence of two chlorines attached to the aromatic ring not only makes this molecule less flammable and more chemically resistant, but it also helps it stand out in extractions during purification. The pyridine ring gives it a certain stubbornness; while not volatile like some compounds, it isn’t as dangerous as strong solvents or acids, but nobody would call it benign, either.
Anyone who manages hazardous materials knows that labels count. The industry defines 2,4-Dichloropyridine by its assay—usually above 98 percent for research grade—and its documented impurity profile. Technical writers emphasize clarity on risks, safe handling, and chemical identity, especially for shipment across borders. Tracking lot numbers and purity isn’t the most exciting part of chemistry, but it’s where the real trust lies: when the flask says “2,4-Dichloropyridine,” a chemist expects that’s what they’re working with. What’s missing is often as important as what’s present—low water content, no unreacted raw materials, and no rogue halogenated quinoline byproducts.
In the lab, few things are as satisfying as seeing a perfectly substituted pyridine in the flask. Most syntheses of 2,4-Dichloropyridine start with a simple pyridine ring, then introduce chlorine atoms stepwise, often relying on proprietary variants of Sandmeyer or direct chlorination routes. Some of the yield-boosting tricks involve controlled temperature, carefully chosen solvents, and the right catalysts—in practice, these variables make or break industrial-scale production. It’s not just about reagents; it’s about keeping the chlorine where you want it, not on the 3 or 5 positions. Each tweak changes yields, costs, and the environmental footprint of the process, which matters even in bulk manufacturing.
Synthesizing a new compound often relies on these halogenated pyridines. In practice, I and colleagues have taken 2,4-Dichloropyridine and run nucleophilic aromatic substitution, swapping out a chlorine for amines or alkoxides. It’s the classic strategy to build active pharmaceutical ingredients, with the pyridine ring serving as a backbone that holds functional groups in the right configuration. The molecule’s structure means it can handle strong bases or nucleophiles, which opens up all kinds of derivative products: active drugs, complex agrochemicals, and molecular probes used in biology. Each interaction reveals new routes for molecular innovation.
The world of organic chemistry language can be a headache for newcomers. 2,4-Dichloropyridine gets called by a range of technical names: sometimes as Pyridine, 2,4-dichloro-, or by its CAS number. In industry, clarity on synonyms is more than academic—one wrong label can lead to wasted hours or even safety issues. I’ve seen the confusion when suppliers and researchers speak across different naming conventions, so a unified chemical language isn’t just for tradition, it’s for getting work done safely and effectively.
Working with 2,4-Dichloropyridine means sticking with the habits that keep people safe: chemical-resistant gloves, goggles, good ventilation, and storage away from incompatible reagents like strong oxidizers. GHS (Globally Harmonized System) pictograms point out health hazards—irritant, harmful if swallowed—but that’s just the start. Training in labs covers what to do with spills and accidental contact, because the real risks don’t always appear in bold on a safety sheet. Chronic exposure brings concerns about organ toxicity, and safe waste disposal keeps labs in compliance. These aren’t just legal hoops; they’re daily practices that protect people and communities near chemical plants or research centers.
2,4-Dichloropyridine plays an unglamorous but crucial role in many modern products ranging from antimalarial drug precursors to herbicides. Agribusiness uses intermediates made from dichloropyridines to make selective weed killers that protect crops and raise yields. In the pharmaceutical sector, a few blockbuster drugs and specialty treatments rely on this molecule for their synthesis. My own experience working with researchers hunting for new anti-infectives included using 2,4-Dichloropyridine as a base to build new compounds faster than before. It doesn’t get headlines, but its fingerprints appear on everything from improved crops to next-generation medicines.
There’s a rush in the chemical industry to improve yield, safety, and sustainability, and 2,4-Dichloropyridine finds itself in the crosshairs. Green chemistry pushes synthesis routes that skip hazardous reagents or cut down on waste. In academic labs, students try to swap old-school methods for catalytic alternatives or less toxic chlorinating agents. Industrial research departments focus on process intensification, not only to save money but to reduce environmental footprints. Tools like flow chemistry and automation can cut reaction times and lower risks—but each advance builds on the reliable backbone that molecules like 2,4-Dichloropyridine provide.
Every promising molecule brings caution. Studies of dichlorinated pyridines, including 2,4-Dichloropyridine, show moderate toxicity profiles and alert regulators and industry watchdogs alike. Acute exposure can lead to skin and respiratory irritation, so most protocols call for careful handling under a hood. Animal studies show specific organ effects at higher doses, raising concern about chronic exposure in manufacturing settings. Environmental impact research follows the breakdown products and how they cycle through soil and water, reminding manufacturers and labs that every intermediate needs tracking from cradle to grave. In real labs, toxicity research means regular blood tests and strict adherence to exposure limits—not because it’s theoretical, but because it affects real people.
Looking ahead, 2,4-Dichloropyridine could shift into greener production lines or inspire new synthetic routes for the next wave of drugs or crop protection tools. Innovation rarely comes from completely new materials; improvements often splice onto the molecules already in the toolkit. Advanced synthetic methods, regulatory pressures, and an eye on end-of-life disposal are shaping how industry and academia approach this compound. Greater demand for specialty chemicals in personalized medicine, new agrochemicals with better safety profiles, and streamlined manufacturing processes could all depend on reliable intermediates like this one, as long as they get produced with a mind toward human health and environmental impact. In practice, it means ongoing vigilance from chemists, tighter safety standards from industry, and honest conversations about the trade-offs that come with powerful chemistry.
The chemical 2,4-Dichloropyridine doesn’t roll off the tongue, but its purpose reaches far into corners of the modern world many people never think about. Anyone who’s spent time looking into how crops get protected from insects or how new medicines come to market has probably brushed past the trail of this chlorinated pyridine. Its value often escapes mainstream attention, but a closer look reveals a story worth noticing.
Years ago, I spent time with a friend who worked in crop science research. Back then, terms like “pyridine derivatives” meant almost nothing to me, but he explained how these kinds of chemicals help shape the backbone of much of what supports our food system. Some herbicides, particularly for grains and certain vegetable crops, depend on building blocks like 2,4-Dichloropyridine. It functions as a key intermediate, forming the stepping stone between simple chemicals and complex molecules that actively safeguard fields. Farmers rarely see this powdery substance, but it’s right there in the origin story of several modern crop protection agents.
The same building block logic applies in pharmaceutical labs. Scientists use 2,4-Dichloropyridine to create other molecules—the real heavy lifters that tackle infections or manage chronic disease. Drug discovery often resembles trial and error, requiring small but important adjustments to chemical structures. This compound provides chemists just the tweak needed to change a molecule’s behavior. Take antivirals or cardiovascular agents: often these medicines start as something basic, then gain real punch through the addition of chlorinated rings just like the one in question.
Having worked in environmental policy circles, I’ve seen the flip side, too. Chemicals bridging the journey from lab to field sometimes spark debate over safety. Questions crop up around manufacturing waste or what spills could mean for soil and water. That’s a real concern—not just for activists, but also for those trying to hold food production steady on this crowded planet. The story of 2,4-Dichloropyridine shows a push and pull between progress and risk management. Mistakes in the past have taught us that ignoring accidental releases or improper disposal can spiral into long-term issues for ecosystems and communities.
Mitigation doesn’t mean halting science. Oversight in the form of tight regulations on handling and transport helps. Facilities benefit from strong internal monitoring and waste treatment technologies. In my own experience, transparent reporting on chemical use builds trust with neighbors who feel the unease of factories nearby. Collaborations between industry, local regulators, and public health experts play a quiet but vital role. They make sure innovation in chemistry leads to real gains—without leaving another set of problems in its wake.
Chemicals like 2,4-Dichloropyridine rarely attract headlines outside of specialist circles, yet their ripple effects reach grocery stores, clinics, and municipal water systems. The trick is staying honest about strengths and limitations. There’s no ignoring how much modern agriculture and medicine draw on these hidden ingredients to keep shelves stocked and treatments available. Yet nobody benefits from shortcuts that ignore spillover harm. If our generation wants to hand down better systems, watchdogs and everyday citizens alike must keep asking: How are these substances managed — and at what cost? Genuine progress means current benefits don’t sow trouble for future neighbors. Keeping that promise turns an obscure chemical into something all of us have a stake in.
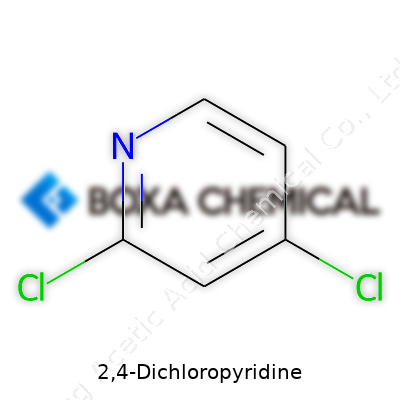
Step into the world of industrial chemistry and often you’ll encounter compounds that have changed farming, pharmaceuticals, and even electronics. 2,4-Dichloropyridine is one of these. Its chemical formula, C5H3Cl2N, lets anyone with a bit of background spot that this is a pyridine ring with two chlorine atoms hooked onto it. That rings a bell for people working with agrochemical building blocks or fine chemicals.
Breaking down that formula: five carbons, three hydrogens, two chlorines, and one nitrogen. The shape of the molecule means it lends itself as a starting material for more complex substances. In the lab, chemists prefer compounds that offer both versatility and reliability. 2,4-Dichloropyridine often finds itself as the go-to when synthesizing drugs or making more advanced chemicals. The two chlorines open doors to further reactions. They can be swapped out, transformed, or used to steer the molecule down different synthetic paths.
Clarity around chemical makeup isn’t just about textbook definitions. Real-world safety and regulatory compliance depend on recognizing exactly what’s on hand. Over the years working in a research environment, I've seen that mistakes in basic identification lead to costly delays—sometimes much worse. Chemical formulas describe more than proportions; they spell out risks, storage needs, and the backbone of any lab protocol. For 2,4-Dichloropyridine, knowing the structure helps with safe handling since dichlorinated aromatics tend to be a touch more persistent and potentially toxic than their single-chlorine cousins.
Compounds like this show up in many synthesis routes for pharmaceuticals and agricultural products. They offer stable platforms for attaching new atoms or groups. That helps speed up innovation but also means release into the environment or mishandling can pose health and ecological problems. Over my time in the field, I've watched as labs implemented tighter inventory checks and waste management policies after minor mishaps escalated. A formula might seem simple, but what it enables can be complex.
Talking with colleagues who manage chemical procurement, the shared focus isn’t just supply; it’s always about safe sourcing and sustainability. Regulations keep tightening around organochlorines due to their tendency to linger in nature. Makers of pharmaceuticals and crop protectants look for cleaner alternatives or tweak existing processes to reduce environmental impact. Many companies now run regular trainings, stressing chemical literacy from the ground up. Even for seasoned scientists, that habit of going back to basics—like writing out formulas—makes a difference.
The formula C5H3Cl2N tells a story of both risk and potential. It shows up in supply requests and safety sheets, setting the foundation for everything that comes after. Some prefer to gloss over details, but keeping sharp with those molecular facts pays off in productivity and safety. It’s not just about ticking boxes or passing inspections; it’s about respect for the power and consequences of these deceptively everyday chemicals.
Anyone who’s spent time in a lab or plant recognizes the need for practical thinking when you’re working with chemicals like 2,4-Dichloropyridine. This isn’t a substance you wave off; its reactivity and the effects on skin and respiratory tissue can’t just be ignored. Workers often want to get the job done fast, sometimes cutting corners, but that’s where trouble finds you. I remember the sharp chemical smell after a dropped bottle during an internship. That crash of glass, the sting in your nose, and the slap of panic stayed with me far longer than the safety briefings did.
Some colleagues still see goggles or gloves as a hassle. But a single splash on unprotected skin and you learn to respect the recommendations. Chemical burns from this compound heal slow and the discomfort messes with concentration. Handling 2,4-Dichloropyridine demands good ventilation. An open window doesn’t do the trick, so real exhaust hoods earn their spot. It’s about keeping vapors away from your lungs—the chronic effects on breathing aren’t worth the risk, something reinforced by long-running case studies and regulatory health reports.
Over the years, automation has helped by taking some of the direct handling out of human hands. Still, automation brings its own dangers. Spills and leaks won’t go away entirely, so experienced operators always keep an eye on seals, gasket conditions, and temperature readings. No automated alarm compares to catching an off-smell or a color change before a problem turns serious. That simple habit of double-checking never gets out of date.
Proper labeling and storage matter, not as a paperwork activity but to avoid cross-contamination. Years back, one mislabeled container led to an unexpected reaction. A small mix-up in the storage rack can snowball. Chemical literacy through hands-on safety drills makes more sense than endless binders full of technical jargon. I’ve seen crews respond faster after practicing spill cleanups with real simulants, not just sitting through videos. It breaks the monotony and makes lessons stick when muscle memory kicks in.
Inhalation is a big issue with volatile compounds. Disposable masks only go so far. Real protection means using properly fitted respirators with the right cartridges for this class of chemicals. Throwing on a paper mask and hoping for the best doesn’t cut it. The long-term price for sloppy habits comes out of your health and, in some cases, reputation. Stories about permanent damage travel down the grapevine for good reason.
One fix that often doesn’t get enough credit is training tied to real incentives. Rewarding teams for zero accidents or optimistic reporting encourages honest feedback, not cover-ups. No one should feel pressured to hide a near-miss; those are the moments that signal a bigger problem brewing. Transparency leads to safer practices and drives a culture that values well-being over quick output.
Regulatory agencies lay out clear guidelines for hazardous materials, and following these shouldn’t just be about avoiding fines. It’s about recognizing that every shortcut means someone, somewhere, could pay for it. Sharing lessons—both good and bad—keeps the cycle of learning steady. In the end, working with 2,4-Dichloropyridine becomes much safer when you combine practical respect for the danger with habits built on first-hand experience and solid teamwork.
Many chemicals have complicated names and even more complicated impacts on human health. 2,4-Dichloropyridine falls into this category. For people who don’t work with chemicals every day, that name may not mean much, but for scientists, factory workers, and environmentalists, it stands out because of its potential risks.
Let’s put it simply: most folks will never run into 2,4-Dichloropyridine unless they’re tied to a chemical plant, laboratory, or agricultural manufacturing. It doesn’t live in over-the-counter products or popular consumer goods. Instead, this compound turns up most often in the guts of industrial production—especially in the making of pharmaceuticals, pesticides, and specialty chemicals.
Working in chemical industries for years, I learned to respect substances like 2,4-Dichloropyridine. Gloves, proper ventilation, and disposal protocols aren’t just for show. Breathing in the dust or getting it on your skin can lead to some serious problems—irritation, headaches, and for some, much worse. According to available studies, exposure at moderate levels can cause respiratory issues, skin inflammation, and eye irritation. Chronic exposure may have more severe effects, though long-term studies haven’t nailed down all the details. There’s enough evidence to make anyone cautious.
It comes down to its chemical structure. The dichloro part means the molecule contains chlorine, which tends to make any organic compound more persistent in the environment. Pyridine rings appear in both helpful drugs and nasty toxins. Chemical experts note that many chlorinated aromatic compounds, once let into the air or water, tend to stick around. Once they’re out there, getting rid of them isn’t cheap or easy.
Animal tests and some cell studies indicate that 2,4-Dichloropyridine can injure the lungs, liver, and kidneys if someone’s exposed above recommended workplace limits. I’ve seen chemical handlers show up at the nurse’s office with rashes, watery eyes, and consistent coughs—all after accidental contact. Regulatory agencies like OSHA and the European Chemicals Agency don’t classify it as a known carcinogen, but their guidelines call for serious respect. Anything powerful enough to irritate your insides deserves a close look.
Imagine solvents and chemical byproducts trickling out of storage barrels. If not managed right, compounds like 2,4-Dichloropyridine leach into soil and water, where they can impact aquatic life. Some studies suggest bioaccumulation could threaten local fish or disrupt small ecosystems. These are not headline-grabbing disasters, but they chip away at biodiversity and food safety.
Standard wastewater treatment doesn’t always break down complex chlorinated compounds. What’s left might slip out the pipe and become everyone’s problem down the river. Many countries have started demanding more thorough pre-release treatment for this reason, but enforcement gaps remain.
No one should try to handle 2,4-Dichloropyridine without training, safety gear, and proper engineering controls. Replacing outdated systems with closed-loop processing, using local exhaust ventilation, and fixing leaks fast all matter just as much as wearing goggles. Facilities doing testing once a year don’t cut it. Workers and their health reps should demand tougher exposure monitoring and practical training—these push managers to prioritize safety, instead of waiting for an inspector to show up.
Innovation could change the story. Safer alternatives for certain uses deserve real investment and attention. Until then, treating 2,4-Dichloropyridine as a potentially toxic chemical—one that sticks around and bites back if ignored—serves everyone’s long-term interests. Factories, researchers, and lawmakers need to keep pressing for stronger protections, because prevention always costs less than cleanup and lost health.
2,4-Dichloropyridine stands out as a crucial chemical for many labs and manufacturing floors, often used for synthesizing pharmaceuticals and other specialty compounds. Experience has taught me that chemicals with reactive chlorines and nitrogen atoms can surprise even seasoned chemists. Hidden risks catch up fast, especially if proper care slips into daily routines. Many hear about accidental exposures or near-misses only after procedures falter. Let’s unpack what safe storage really looks like and why a few basics make lasting differences for both workers and the environment.
This compound's toxic nature deserves respect. Exposure through inhalation or skin contact leads to irritation or worse, especially during spills or poor transfers. 2,4-Dichloropyridine isn’t something anyone wants near food or open drink containers. My own lab days taught me that even quick access to a chemical can tempt sloppy shortcuts if oversight lags. Once, one bottle ended up too close to an open flask, and the headache that followed cost hours of work and risked health. Handling it with proper gloves, goggles, and lab coats should never be seen as overkill.
Safe storage begins by separating strong acids, bases, or reducers, as this chemical may react and form hazardous vapors or byproducts. I've watched new staff assume that sealed caps fix every storage problem. They don’t. Such confidence leads to headaches — literally and figuratively — down the road. Segregation on designated shelves goes much further than mere habit. One real solution: Use secondary containment, like plastic trays. These trap leaks, making small mistakes manageable, and keep containers stable if someone accidentally bumps a shelf.
Many discoveries in chemistry started with temperature mishaps, and 2,4-Dichloropyridine has no exception here. Store it in a cool, dry space and keep it away from direct sunlight or wet rooms. Water gets into containers, degrades the compound, and changes how it behaves in reactions. Humidity swings speed up container corrosion too. Keeping every bottle tightly sealed preserves not only freshness, but also ensures fumes don’t escape to create sneaky hazards nobody wanted to see.
Labels make or break good storage systems. Faded or vague labels put everyone at risk, especially for newcomers or when work gets busy. I’ve seen workers pour the wrong substance because a marker wore off. Always mark original labels with the date received and date opened, as shelf life matters more than many expect. Rotating older stock to the front ensures nothing gets forgotten for years at the back of a cabinet.
Good storage habits for 2,4-Dichloropyridine grow from daily vigilance, not grand policies. Avoid overstocking. Chemicals that gather dust often end up forgotten, increasing the chance that someone mishandles them later. Regular audits — just simple walkthroughs — help spot outdated bottles, corroded caps, or misplaced items. Peer checks have caught long-forgotten vials more than once in labs I’ve worked in.
Careful storage balances safety, work efficiency, and environmental risks. Every time this chemical gets tucked away correctly, it protects both those in the room and the wider world outside. The real win comes from daily commitment and fresh eyes, not forgetting that small habits lead to bigger safety gains over time.