The tale of 2,6-Dichlorobenzonitrile runs parallel to the steady drumbeat of twentieth-century chemical advancement. By mid-century, curiosity about halogenated aromatic compounds pushed researchers to look at these molecules with a sharper gaze. Many discoveries came from academic and industrial collaborations, often motivated by demand from agriculture and specialty manufacturing. The chrysanthemum fields and postwar chemical labs fed each other’s needs. Then, as environmental regulation came into sharper focus, interest shifted from basic synthesis to impact, safety, and environmental persistence. Many of us working in analysis during the eighties and nineties ran across 2,6-Dichlorobenzonitrile as a molecule that kept showing up—not just in research but also in regulatory paperwork and industry seminars discussing responsible production and disposal.
2,6-Dichlorobenzonitrile holds its place as an intermediate, most familiar to those in agricultural chemistry. The molecule captures attention not only for its selective bioactivity but also for its role in developing downstream products where precision matters. Any chemist who has spent time in synthesis labs has probably encountered it as a starting block for crafting more complex structures. Its persistent appearance on shipment manifests shows that it remains a workhorse, despite changes in regulations and market trends.
Solid at room temperature, 2,6-Dichlorobenzonitrile forms colorless or off-white crystals, giving little away to the eye but promising subtle power in combination. Its relatively high melting point, noticeable odor, and moderate volatility require attention in handling, especially in an area with inadequate airflow. Chemically, the presence of both nitrile and chlorinated aromatic groups makes it stable under standard conditions yet reactive enough for further transformations in the right setting. Many chemists find these balancing acts between stability and reactivity handy for designing robust organic synthetic routes.
Significant precision goes into labeling and qualifying batches, driven partly by evolving transport and workplace regulations. Purity claims above 98 percent serve as a starting point; tighter specs help keep liability at bay. Packaging standards reflect not only regulatory codes but also basic common sense, drawing on experience in chemical logistics—sealed drums avoid contamination and ensure safety for workers. Reliable traceability stands as a must, not just for compliance auditors but for on-the-ground troubleshooting if a batch ever throws up problems during downstream processing.
The synthesis of 2,6-Dichlorobenzonitrile often draws from chlorination of benzonitrile under controlled conditions or dehydration of suitable chloro-substituted benzyl derivatives. Choosing the right method depends on access to intermediates and disposal options for any byproducts. While handling strong halogenating agents or nitrile-precursors, real challenges show up in controlling reaction temperature and quenching unwanted side products. I remember spending long hours calibrating feed rates, always aware that an uncontrolled exotherm could undo good planning—not to mention the mess left on the fume hood floor. For small-scale research labs and industrial facilities alike, process control systems and experienced operators make the difference between efficient production and unexpected downtime.
The molecule’s reactive sites open the door to further functionalization. Our own group devoted weeks to experimenting with coupling reactions, discovering that targeted reductions sometimes yield amines useful in pharmaceutical development. Success in these ventures often stems from patience and close monitoring—small tweaks in reagent ratios and temperature profiles frequently produce substantial shifts in byproduct formation and yield. Chlorinated aromatics do not forgive impatience or shortcuts, and safety protocols exist for good reason; people learn quickly, sometimes the hard way, that skipping steps in quenching or extraction can turn a routine job into a hazardous situation.
Chemists often encounter the same compound under a variety of synonyms: 2,6-DCBN, dichlobenil, or more systematic identifiers based on IUPAC nomenclature. Reviewing regulatory documents, it becomes clear these names can cause headaches, especially during international collaborations. Cross-referencing synonyms while negotiating import permissions or project documentation has always felt less like paperwork and more like detective work. Precise documentation avoids shipment delays and prevents confusion among partners or regulatory agencies trying to track inventories, especially in multi-national settings.
Handling 2,6-Dichlorobenzonitrile requires careful attention to both manufacturer guidance and common-sense lab safety. Respiratory protection for powder handling, reliable dust extraction systems, and gloves form the everyday toolkit for users, whether they face kilograms or grams. I recall annual safety audits as an opportunity to swap lessons with younger colleagues and spot bad habits slipping into routines. Regulations enforce minimums, but culture and shared practice go further—unannounced spot checks and peer observations wield more influence on long-term safety than postings on breakroom walls. Emergency planning drills reinforce the message that hazardous chemicals demand constant attention; accidents often sneak in during the complacency that grows from routine.
A significant chunk of the global production ends up in herbicide formulations, valued in agriculture for selective weed suppression. Some colleagues in crop sciences recall the transformation wrought by chemical weed control, blending technological optimism with a deep sense of responsibility toward unintended environmental consequences. In fine chemicals, the compound serves as a stepping-stone, contributing to flavor, fragrance, and even dye manufacturing. Academic labs appreciate its role in methodology development, while regulatory specialists track environmental residues in water and soil, watching for patterns that could shape the debate about continued agricultural use.
Teams pushing forward on greener chemistry have kicked off efforts to produce 2,6-Dichlorobenzonitrile through less wasteful or hazardous routes. Some work on engineered biocatalysts aims to bypass harsh reagents and dirty byproducts. My own experience suggests change moves slowly, especially when commercial-scale infrastructure already favors established processes. Real progress grows out of partnerships between researchers, process engineers, and companies willing to prioritize sustainability over short-term cost savings. Funding for this kind of work rarely pours in, but curiosity and regulatory pressure can shake loose new ideas and grant money. Conferences where plant managers talk shop with academic project leaders show what collaboration looks like—the best solutions rarely come from one side alone.
Long-term toxicity studies shine a spotlight on environmental persistence and bioaccumulation. Animal testing and environmental fate models trace the life cycle from factory floor to field runoff and, eventually, aquatic organisms downstream. The years after initial launch brought lawsuits, debate, and tighter controls after evidence showed environmental exposure affected non-target species. Industry responses range from product reformulation to improved containment technology and real-world monitoring. For those working in labs or regulatory bodies, the priority focuses on controlling worker exposure and keeping the compound out of environments not intended to receive it. Medical monitoring of exposed workers provides feedback, helping steer policy toward ever-safer workspaces.
In conversations with researchers and industry planners, realistic hopes for 2,6-Dichlorobenzonitrile now orbit around sustainability, environmental compatibility, and regulatory adaptation. Tighter emissions regulations and moves toward precision agriculture could shift the mix of applications, driving more focused use. The compound will stick around as a research tool and building block, but product stewardship and green manufacturing stand ready for stronger emphasis. Training chemists and plant operators in holistic lifecycle analysis promises a meaningful step forward. Funding for cleanup chemistry and in-situ remediation stands to make a real impact, as long as stakeholders push for solutions that acknowledge both risk and reward over years, not quarters.
Talk with anyone who’s worked in agriculture or gardens on the edge of farmland, and you’ll hear a lot about weeds. People pour hours into pulling them out or fighting them off, because those little green invaders turn crops and lawns into daily struggles. Chemical solutions make their way into the conversation, and that’s where 2,6-dichlorobenzonitrile gets a mention—not as a household name, but as a piece of the puzzle for controlling unwanted growth.
2,6-Dichlorobenzonitrile goes by an even trickier name in practice: DCBN. In the real world, its most common application comes as an active ingredient in selective herbicides. For years, this compound found its way onto fields, especially where carrots, onions, and other root crops grow. The reason farmers and big growers turned to DCBN stems from its knack for knocking out tough weeds without hurting certain crops. DCBN works by tinkering with the way weed seedlings grow, disrupting their roots and stopping them before they get any traction in the soil. The result is a cleaner harvest with less back-breaking labor.
The appeal of DCBN isn’t just about convenience; it's about efficiency and crop yield. When you lose fewer crops to weeds, you spend less money and get more to market. With the increasing pressure around the world to use land efficiently and squeeze every bit of food from shrinking acres, DCBN promised a practical answer. Some reports pointed to better harvest numbers, less erosion thanks to minimal tillage, and more predictable results in fields battered by invasive plants.
The other side of the story always deserves mention. As a long-time gardener and someone who grew up around small farms, I remember warnings about herbicide drift and “odd patches” where nothing grew the next season. Chemicals that kill weeds may threaten other plants or soil microbes. DCBN isn’t immune to this criticism. Studies flagged its persistence in soil and the potential to harm aquatic life if it washed away in runoff after heavy rains. People who live near sprayed fields sometimes worry about exposure, since many herbicides get carried on the wind or in water.
Consumers now ask more questions—how safe is the food, what’s the risk to the community, and does the benefit outweigh the cost? In several regions, regulators put tighter controls around DCBN’s use. Some countries limit or ban it entirely, citing environmental and health risks. Others still allow it but demand strict handling and guidelines to keep residues as low as possible in both crops and water sources.
Clear communication and transparency help. Farmers can keep records of application, share data about alternative cropping systems, and experiment with integrated weed management. Strategies like mechanical weeding, cover cropping, and shifting to new crop rotations can reduce chemical reliance. Researchers continue to study how long DCBN stays in soil and what organisms feel its effects most. Transparency in research builds trust and gives both farmers and the public more say in how tools like DCBN get used.
It’s not enough to look at a chemical just for its benefits. We need regular reviews, new studies, and honest talk about where it helps and where it harms. An older herbicide like 2,6-dichlorobenzonitrile has a footprint that stretches from the field to the kitchen table. It's worth knowing what goes on the crops, how it moves in the environment, and what steps lead to better food and cleaner land. Only by looking at the broader community and environment can we make decisions that balance productivity and safety.
I spent a few years working in landscaping, often tasked with pulling weeds in playgrounds or parks. One day, the foreman rolled in early, warning us to steer clear of the patch sprayed with a chemical weed killer a few hours prior. The sharp, almost bitter smell pricked my nose. That patch turned out to be recently treated with a compound called 2,6-Dichlorobenzonitrile. It’s an ingredient that’s popped up again and again on farm reports and in gardening supply discussions. Sitting at the crossroads of agriculture and industry, this stuff raises big questions: just how risky is it for human health?
2,6-Dichlorobenzonitrile works as a pre-emergent herbicide, keeping weeds from sprouting. It gets poured or sprayed on soil, where seeds absorb it and stop growing. Farmers and groundskeepers like its efficiency because it keeps lazy mornings a little less sweaty. It’s less famous than glyphosate or paraquat, but it’s still found on fields, orchards, and along railways.
My first real worry started with a simple fact: this chemical doesn’t quickly break down. That means it hangs around soil or water for weeks, maybe months. A few toxicological studies logged its persistence, showing it doesn’t degrade fast under sunlight or rain. And when chemicals linger, long-term exposure becomes much more likely. Food chain buildup concerns creep in, especially for folks whose jobs regularly bring them near treated areas.
From the health angle, skin contact and inhalation sit at the front of the risk line. 2,6-Dichlorobenzonitrile can irritate eyes, nose, and skin. It feels like a sting or burn for some, and prolonged exposure has caused rashes and breathing difficulty in agricultural workers. The EPA and European regulatory agencies both marked it as “harmful if swallowed or inhaled,” which isn’t just a legal warning—it reflects what poison centers have seen in real-world cases.
Lab tests in animals point to much bigger risks. Mice fed significant doses developed organ changes, especially in the liver. Inhalation exposure in studies brought on respiratory tract effects. Long-term or high-level exposure sometimes tipped the scales toward more serious outcomes, such as developmental or reproductive issues. There’s limited data from human studies. Still, most toxicologists argue that the animal data offers enough reason to keep it out of our lungs and off our skin.
Packing the shelves with personal protective equipment always felt tedious, but during spraying days, it felt absolutely necessary. Gloves, masks, and long sleeves kept symptoms at bay for me and my crew. Looking at broader solutions, more routine monitoring of residues in water and food helps spot contamination early. The real wins show up when companies swap out persistent chemicals for ones that degrade safely or use non-chemical weed control.
Beyond equipment, clear safety training stands as one of the best lines of defense for those working with agricultural chemicals. If a town or farm wants to protect its laborers, that means more than memo printouts—it means hands-on demos and refresher courses. Consumer pressure also helps. Stores can choose to stock less hazardous options and label more clearly, so shoppers figure out what’s risky and what’s not without confusion.
Everyone benefits from good, local data on pesticides and their health impacts. Researchers and health advocates need backing to track these chemicals in actual communities, not just lab mice. Farmers, groundskeepers, and parents all deserve questions answered with clear, solid evidence.
A lot of folks wonder why storage rules for chemicals like 2,6-dichlorobenzonitrile seem overly strict. It’s easy to think of these rules as red tape until you realize how quickly things can go sideways. My background working in a research lab taught me to treat every substance—especially ones with chlorine and nitrile groups—with real respect.
2,6-dichlorobenzonitrile often pops up in herbicide formulations. Farmers, warehouse staff, and distributors handle it regularly, so keeping storage easy and safe matters. Cases of accidental exposure rarely make the news, but anyone who got a whiff of the fumes or felt the stuff on bare skin remembers it long after. Even minor leaks cost time and money, trigger health risks, and damage trust in safety routines.
Chemical stability depends a lot on temperature. Heat encourages breakdown and sometimes sparks dangerous reactions. That’s not a risk you want in a storeroom. I’ve watched thermal cycling—hot days followed by cool nights—stress packaging and cause liners to degrade. The simplest fix is climate control. Most industrial guides recommend cool, dry storerooms. Keeping the temperature consistent not only preserves product quality but also protects building occupants.
Moisture deserves respect, too. 2,6-dichlorobenzonitrile should always stay bone dry. Even trace water can promote undesired hydrolysis, since chlorinated aromatics don't typically play nice in wet conditions. Water can corrode drums and packaging, which ruins labels and eventually causes leaks. Stacking containers on pallets instead of concrete floors, and avoiding storage near wash stations or humidifiers, limits these risks considerably.
There’s a myth that only highly volatile substances need fresh air flow. Anyone who has ever cleaned up after a leaky bag knows chemical vapors build up fast. Good ventilation clears out anything that escapes and keeps work areas much safer. There’s science backing this—studies show well-ventilated spaces see lower rates of exposure incidents with aromatic nitriles. Opening windows isn’t good enough in industrial spaces. Ventilation systems that don’t recirculate air make the biggest difference for worker health.
Mixing chemicals together in storage sounds reckless, but it still happens. Combining acids or strong bases with chlorinated organics can spark serious trouble. I’ve seen sulfuric acid stored right next to organic chemicals in older warehouses, and staff had no idea about the risk. Training—actually walking through the rules with employees, not just handing out a binder—closes this gap better than any poster. Banning food and drink in storerooms, labeling containers clearly, and using secondary containment trays stops cross-contamination and limits exposure during spills.
Accidents rarely make for exciting headlines, but everyday safety lapses can stack up fast. OSHA and similar agencies regularly report citations for storage violations, and most are preventable. Smart storage—real temperature control, dry conditions, good airflow, segregation, and practical training—does more than tick boxes. It actually protects people. Think of it as building trust in every link of the supply chain. That’s a lesson I learned on the job, not from a textbook.
You can’t sugarcoat the risks tied to 2,6-Dichlorobenzonitrile. This chemical works as a potent herbicide and intermediate in manufacturing, valued for its role in keeping many sectors ticking along. Anyone who’s spent days in labs or industrial plants knows a chemical with “chlorine” and “nitrile” together will raise eyebrows and call for focus.
Stories circulate among workers about how careless splashes or dusty air ended in hasty eye rinses or hives. This isn’t some relic from an old-time plant; even today, poor handling sends people to medical care. Research links 2,6-Dichlorobenzonitrile exposure to skin, eye, and respiratory irritation, with animal studies raising flags over organ and nervous system damage after repeat contact. I’ve seen folks underestimate low-volume spills, skipping gloves to “just wipe up quickly,” only to regret it with raw skin hours later.
A culture of cutting corners often puts pressure on workers to skip simple steps like pulling on goggles or double-checking gloves. Safety gear sometimes gets dismissed as an inconvenience, especially for quick jobs, but a day lost to chemical burns or persistent coughs has a way of changing minds. Chemical-resistant gloves, protective eyewear, and lab coats aren’t luxuries; they’re the real dividing line between a routine day and a health scare. Proper ventilation keeps vapors and dust out of lungs—never optional with this compound.
Ask custodial staff or technicians how they feel after dealing with open containers, and most will mention headaches or nausea. 2,6-Dichlorobenzonitrile gives off fumes that linger if containers aren’t tightly sealed. Where it’s stored can’t be an afterthought either: dry, temperate spaces well away from acids or anything flammable matter more than a sign on the door. For clean-ups, dry sweeping stirs up clouds nobody wants to breathe—wet mops or vacuum systems with filters work far better at trapping residue and dust.
I recall a time a new hire dove straight into measuring without checking the chemical’s SDS, thinking the routine gloves from a previous job would offer solid protection. More time spent on realistic, example-driven safety training—pointing out not just the rules but the real stories—prepares people better than any checklist. Regular refreshers keep hazards fresh in mind, especially after close calls that get brushed under the rug.
No single rule slashes all risk, but companies can keep genuine dialogue open among front-line staff. Input from the people who work hands-on with 2,6-Dichlorobenzonitrile often leads to the most sensible improvements, whether it’s better air filtration or reminders posted where spills happened before. Industry guidelines and legal requirements help, but it’s regular folks watching out for each other who create a culture where careful handling becomes second nature.
Anyone handling strong chemicals knows caution has a short shelf-life if leadership chases efficiency over health. Taking shortcuts on safeguards always catches up. If an environment expects workers to “tough it out,” risk only climbs higher. Investing in sturdy personal protective equipment and honest safety talks holds more value than any quarterly report or day saved on a rushed job. In the end, respect for 2,6-Dichlorobenzonitrile isn’t about fear—it’s about understanding what’s on the table and valuing everyone’s right to a safe shift and a healthy life.
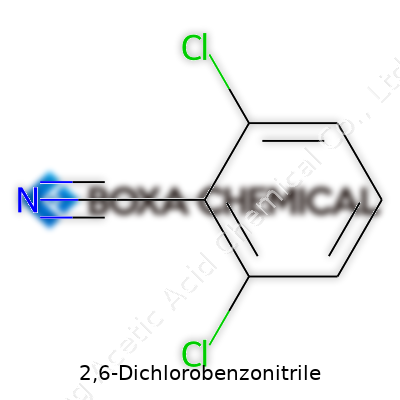
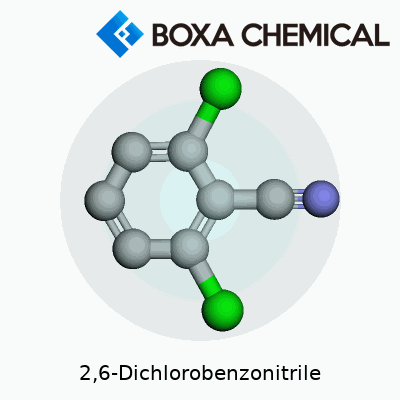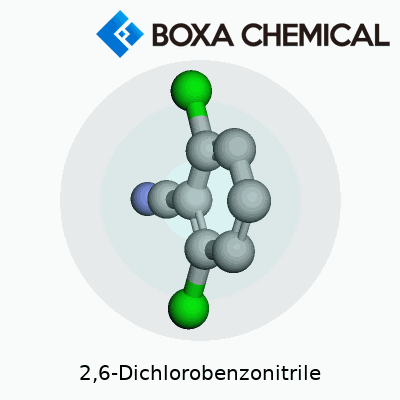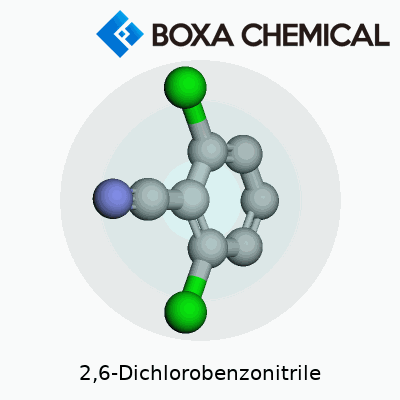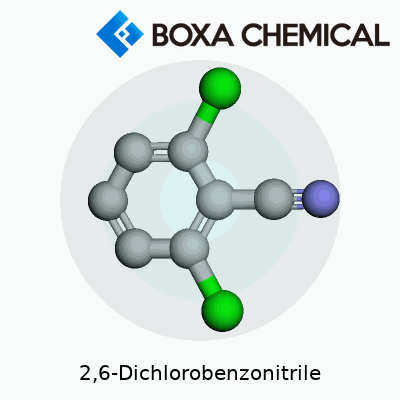
2,6-Dichlorobenzonitrile doesn’t win any beauty contests in the molecular world, but its architecture is easy enough to picture if you know a bit of chemistry. The skeleton is made from a benzene ring, that flat, six-sided carbon loop kids usually see in high school textbooks. Here, two chlorine atoms attach themselves at the second and sixth positions on the ring. The structure gets more character with a nitrile group (–C≡N) fixed at position one. This mix of chlorine and nitrile glued to the benzene ring really changes how the molecule behaves.
Count the atoms, and you find that the molecular formula shakes out to C7H3Cl2N. Seven carbons, three hydrogens, two chlorines, and a nitrogen—pretty lean for a chemical that shows up in herbicide discussions and laboratory benches everywhere.
It’s not just chemical curiosity that pushes people to map out molecules like 2,6-Dichlorobenzonitrile. In my time working with pest management researchers, I saw this compound pop up in debates about environmental safety. The structure tells researchers how a chemical will behave around soil, water, and living things. The placement of those chlorine atoms makes it tougher for bacteria to break the molecule down in nature, which influences both its persistence and the potential risks to surrounding ecosystems.
Regulatory agencies use this information to gauge whether a chemical is safe to use or needs tighter restrictions. You can’t talk about herbicides without mentioning environmental impact, and 2,6-Dichlorobenzonitrile’s structure makes it more stubborn against decomposition. That means it can hang around in the environment a while after application, affecting not just weeds but sometimes organisms far outside its original target.
This stickiness in the environment brings up real questions about how we use chemicals in agriculture and land management. In past discussions with farm advisors, I heard a common worry: If something lingers, it could end up in places you don’t want—drinking water, non-target plants, or even food supplies. Studies have shown that some derivatives of benzonitrile compounds, especially those carrying chlorine, can accumulate and work their way into food chains. That doesn’t always mean disaster, but it urges caution.
A smarter approach starts by encouraging more targeted use. Application methods matter. Precision spraying and strict timing help minimize leftovers. I’ve seen farms switch to integrated weed management—mixing physical removal, crop rotation, and lower doses of the right chemical. These strategies sharply curb environmental loading. Supporting research efforts to break down persistent compounds with soil-friendly bacteria or low-impact sunlight-based reactions also draws a lot of promise. Some labs are mapping pathways for natural degradation even now, using the unique structure of molecules like this one as a clue.
All this comes down to making smarter decisions. Understanding the nuts and bolts of 2,6-Dichlorobenzonitrile’s structure ensures safety calls are based on science and experience, not just marketing. On the ground, farmers, scientists, and regulators lean on that knowledge to protect soil, water, and food from unwanted leftovers, proving chemistry isn’t just for the lab—it shapes the world beyond.