Walking through the history of chemical synthesis, 2,6-dichloropyridine stands as one of those compounds that rarely grab headlines yet consistently power progress. Chemists have worked with derivatives of pyridine for over a century, inspired by their utility in both medicines and the fields that feed us. Back in the mid-1900s, when industrial production scaled up, chlorinated pyridines became building blocks for a wave of new pharmaceuticals and crop protection agents. Generations of researchers have learned, sometimes the hard way, how to coax more selectivity and control from each synthetic step. Over decades, the focus shifted from batchwise, low-yield lab processes to robust industrial routes. Each improvement layered chemistry learns from those who wrestled with challenging halogenations or struggled to separate pesky byproducts. Modern chemists have the benefit of a paper trail fraught with frustrated notes, breakthroughs, half-failed reactions — and every improvement owes a debt to those old experiment logs.
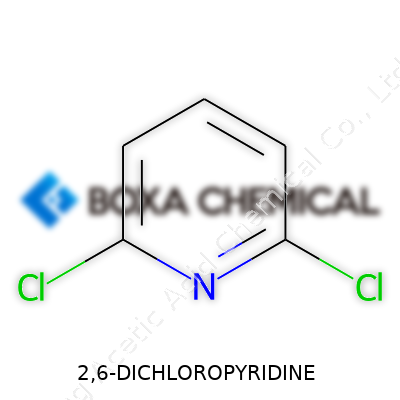
2,6-Dichloropyridine is no household name, but its fingerprints show up in everyday materials and medicines. It's a small molecule with two chlorine atoms locked in at the second and sixth positions of a pyridine ring. This particular pattern of chlorination lends itself to versatility — acting as a scaffold in agrochemical synthesis, cropping up in fine chemicals, and laying the groundwork for advanced pharmaceutical intermediates. Most people outside the chemistry world wouldn’t recognize it, yet if you trace upstream supply chains for crop protection or certain medicines, you’ll find this compound quietly at work.
2,6-Dichloropyridine tends to be a solid at room conditions, often appearing as a white or off-white crystalline substance. Noted for its distinct odor, it dissolves in organic solvents while showing minimal solubility in water. This solvent compatibility and the electronic influence of the chlorine atoms make the molecule more reactive than simple pyridines. Reactivity, here, means the ability of this compound to serve as a handle for more advanced modifications. Its melting and boiling points, along with density and vapor pressure, stay in a range that feels familiar to anyone who’s handled small halogenated aromatic compounds.
From an operational viewpoint, buyers expect strict assurances on purity and consistency. Quality control labs frequently rely on gas chromatography or mass spectrometry to check for residual starting materials or byproducts. Material intended for pharmaceutical synthesis must meet even tighter specifications, often rated upwards of 98% purity. During research projects, impurities as low as a fraction of a percent can trip up downstream reactions, leading to hours of troubleshooting. On bottles and bags, the labeling spells out hazards, purity, and batch number. Such details may feel like bureaucracy to some, but anyone who’s lost a week due to mislabeled reagents learns quick respect for detailed tracking.
Most commercial production routes draw from classical chlorination techniques. Pyridine, itself a relatively ubiquitous starting block, can be subjected to directed chlorination using phosphorus pentachloride, thionyl chloride, or similar chlorinating agents. The challenge comes from selectively introducing both chlorines to the right positions without creating a mess of other products. Over time, process chemists have dialed in reaction temperatures, solvent choices, and reagent stoichiometry to favor the 2,6-dichloro substitution. Big manufacturers favor methods that give high selectivity and avoid wasteful over-chlorination or the use of hard-to-handle reagents. Inevitably, tweaks and modifications show up — sometimes out of necessity, sometimes out of fierce competition to cut costs.
Experienced synthetic chemists appreciate 2,6-dichloropyridine for what it offers — two reactive chlorines sitting opposite each other, just waiting for something more interesting. Nucleophilic aromatic substitution lies at the heart of its utility, especially when building more complex molecules for agriculture or medicine. By swapping out one or both chlorines in carefully controlled reactions, researchers unlock whole suites of derivatives. These modifications open doors to more targeted pesticides or drug candidates. On the lab bench, each transformation brings its own fingerprint and set of headaches. Moisture, heat, base, or subtle changes in process can turn a straightforward swap into a prolonged troubleshooting session. Still, the backbone remains favored by countless chemists for its reliability and the breadth of options it provides.
Dig around for 2,6-dichloropyridine in the literature, and you’ll find it wearing several hats. Names like 2,6-dichloropyridin, 2,6-dichlor-1-azabenzene, and systematic numbers like CAS 2402-78-0 appear. Commercial catalogs use these synonymously, though serious researchers double-check to make sure what’s on offer matches the reaction they want to run. Through decades, this variety of names hasn’t stopped the chemical from showing up again and again as a reliable intermediate.
Working safely with 2,6-dichloropyridine means more than throwing on gloves and goggles. Chlorinated organics bring risks by inhalation, contact, and sometimes by accumulation in workspaces or waste streams. Modern best practices draw from hard-won lessons in occupational safety. Well-ventilated hoods, splash mitigation, and clear labeling — all matter. Regular air monitoring reduces exposure risk, and spill management plans matter for anyone who works late and tired. Chemists, engineers, and safety officers teach these steps for good reason: a close call, or worse, can bring a career’s momentum to a halt. Upstream, responsible producers share technical guidance and safety data sheets, putting worker health and environmental protection above mere compliance.
As a cornerstone intermediate, 2,6-dichloropyridine serves roles in more industries than most realize. It crops up as a building block for agrochemical actives — those essential agents that keep crops viable by controlling pests and weeds. Fine chemical synthesis relies on its reactivity to produce specialized ingredients, from flavors to photoactive agents. In pharmaceuticals, its structure is a frequent starting point for making candidate drugs that leverage the pyridine ring’s bioactivity. Development teams weigh both cost and reactivity when choosing intermediates, and this one lands on lists when reliability and versatility matter. In academic projects and industrial setups, its utility keeps it stocked in storerooms where real work gets done.
Research groups and forward-looking companies push boundaries by trying new functionalizations of the pyridine ring. Each year, chemists publish studies exploring faster, cleaner, or more selective routes to key intermediates using this platform. Catalysis research pushes reactivity even further, sometimes unlocking reactions that used to take harsh conditions and yielding new combinations. Analysts and environmental chemists investigate the compound’s fate in soil and waterways, hunting for safer, more degradable substitutes. Others aim for greener process chemistry — solvent minimization, safer reagents, and energy savings. Work in these areas rarely slows because the demand never goes away.
Experiments and real-world data both feed into a growing understanding of chlorinated pyridines and their long-term effects. While acute toxicity for 2,6-dichloropyridine stays in the moderate range compared to more notorious compounds, cautious chemists don’t let their guard down. Studies in rodents and aquatic systems guide regulatory advisories. Waste management teams design protocols to keep run-off and vapors out of reach of people and the environment. For labs, it’s good science and good citizenship to minimize exposure and waste, even when toxic thresholds seem distant during routine work. Regulations and worker training continue to evolve, reflecting both new knowledge and the willingness of the chemical community to put safety over mere productivity.
Looking ahead, the role of 2,6-dichloropyridine won’t fade. Chemistry’s future thrives not only on new discoveries, but on making the most of proven tools. As demand for sustainable manufacturing grows, the industry continues to refine both starting materials and technology. Greener chlorination, careful recovery of spent reagents, and open data on environmental impact will all shape future handling. Researchers explore bio-derived sources, streamline workflows, and chase processes with less waste. For those in pharmaceuticals and advanced materials, this compound’s adaptability keeps it relevant, turning yesterday’s workhorse into tomorrow’s springboard. Chemists set their sights on safer processes, better downstream degradation, and innovative end-use cases. The conversation around 2,6-dichloropyridine remains lively, and real progress hinges on the willingness to push for change without giving up hard-won advantages.
People don’t talk much about molecules like 2,6-Dichloropyridine outside of research labs or industrial plants. You hear “chemical,” and most of us imagine rows of sealed drums in some faraway facility. But this one pops up in places that matter: agriculture, medicine, and material science. I once stumbled across its name scribbled in a dog-eared notebook, long before I realized how many lives get touched by the decisions that go into making compounds like this.
Farmers fight a constant battle with weeds and pests. Pesticides help them stay ahead. 2,6-Dichloropyridine doesn’t end up on your dinner plate, but it plays a quiet role as a building block. Chemists blend it into the recipes for bigger, more complex molecules—the kinds used in top-selling pesticides. Picture the effort behind a successful harvest: scientists one step, growers the other. Without ingredients like this, you’d see empty shelves in the produce aisle after a bad season.
I know someone who works in pharmaceutical development. She once said, “Breakthrough drugs often depend on the smallest building blocks.” 2,6-Dichloropyridine counts as one of those small but mighty pieces. Pharmaceutical teams use it as a starting material while developing certain medicines, especially in cancer and central nervous system research. What matters isn’t just that the drug works, but that it gets made safely and cleanly. As more folks need newer therapies, these chemicals end up backstage, unseen yet essential.
Life changes the minute you admit science isn’t only about test tubes and textbooks. If you work in electronics or polymers, you see these raw materials show up in unexpected ways. 2,6-Dichloropyridine helps build specialty plastics and custom coatings. Sometimes it’s about durability, sometimes about meeting environmental targets with cleaner or more recyclable products. These aren’t glamorous details, but they shape what manufacturers can build for real-world use. From lightweight materials in cars to protective layers on devices, tiny pieces like this one keep the gears turning.
Anyone handling chemicals has stories about close calls and careful procedures. With 2,6-Dichloropyridine in an industrial setting, technicians run strict protocols. Eyes water from a whiff; gloves and masks become routine. Regulations have teeth for good reason. Overexposure risks exist for both workers and the community, demanding proper storage and handling. Environmental regulators step in if companies cut corners, and that’s a protection worth supporting. Transparency—what gets used, how releases get monitored—builds trust and keeps things honest.
No compound stands alone—we rely on supply chains that span countries and standards that cross languages. Companies buying or selling 2,6-Dichloropyridine must audit suppliers closely and support workers with training. Researchers dig into greener, less hazardous alternatives when new risks emerge. Customers and communities can press companies for accountability, whether through local watchdog groups or smarter purchasing choices. Public data about chemical safety should live where everyone can find it. 2,6-Dichloropyridine could be a doorway to progress or a point of concern, depending on how people treat that responsibility. I’d argue that paying attention—demanding transparency and prioritizing safety—drives technology in ways that benefit us all.
Chemicals have a way of driving innovation, but it’s easy to gloss over the fine print—like names, formulas, and weights—when you’re not deeply in the lab. Take 2,6-Dichloropyridine. Its chemical formula is C5H3Cl2N, and the molecular weight comes in at about 162.99 grams per mole. At a glance, these numbers don’t shout their importance, but peel back a layer, and you see why structure in chemistry deserves everyday attention.
Different arrangements of the same pieces will change the story of a molecule entirely. In 2,6-Dichloropyridine, two chlorine atoms clamp onto a pyridine ring at the 2 and 6 spots—sounds dry, but that little tweak gives scientists a building block for designing medicines or pesticides. If you swap out the position of the chlorines, you don’t have the same effects or possibilities. That formula, C5H3Cl2N, makes the difference between a tool and a dead end in synthesis.
At just under 163 grams per mole, 2,6-Dichloropyridine lands in the territory of simple, versatile molecules. Calculating how much chemical you need for a reaction becomes much easier with that number fixed in mind. For labs, there’s no shortcut around precise measurements. Whether you’re mixing a few grams or running a big batch, that data saves money, time, and reduces waste. It’s a real and constant issue for research organizations and industry producers who want to avoid resource-intensive trial and error. Sometimes, a half-gram miscalculation spins into lost weeks.
Mistakes in chemical math aren’t just about wasted material, though. The bigger picture connects straight to safer handling and sustainability. Proper formulas and weights let chemists plan for clean processes, minimize hazardous byproducts, and scale up only what’s necessary. It isn’t glamorous to talk about weighing powder or calculating atoms, but those actions can help cut dangerous chemicals from winding up downstream in water or soil. With stricter environmental rules entering the conversation, getting the basics right forms the backbone for responsible lab work.
More open access to accurate chemical data serves not just those wearing lab coats. Startups, small research teams, and even students benefit from not having to chase down the basics like molecular formula or weight. Information bottlenecks slow down progress, and sometimes misinformation about simple facts leads to costly setbacks.
I’ve seen undergraduate classes get tripped up by a missing subscript or an outdated figure in a database. It isn’t a small hiccup if it shapes how a whole group learns to approach chemistry. Accuracy matters from day one, whether you’re training future scientists or setting quality benchmarks inside a pharmaceutical company.
Putting the chemical formula and molecular weight front and center is more than housekeeping—it’s the start of smarter, safer, and cleaner science. There’s a lesson here for other technical fields: don’t skip over the details that unlock creativity and safety down the line.
Chemists, lab workers, and safety officers know there's no shortcut to proper chemical storage—especially with specialty intermediates like 2,6-Dichloropyridine. This compound pops up in agrochemical and pharmaceutical labs, with its own quirks and risks. The safety data tells part of the story. My own years working around organic halides have taught me: getting this right isn’t just about ticking off legal boxes or obeying a checklist. Lives and jobs depend on it.
2,6-Dichloropyridine gives off a sharp odor and the dust can irritate skin, eyes, and airways. It won’t catch fire as easily as some solvents, but ignoring the potential for harmful fumes or accidental spills has a way of backfiring fast. I’ve seen what happens when people let their guard down; there’s always a price to pay down the line.
This compound handles best in dry, cool spaces. Excess moisture spells trouble, sometimes causing slow decomposition or making small leaks turn into messy problems. I’ve always trusted big, well-marked cabinets with solid air circulation for this sort of material. Stick to 15–25°C if possible, and keep the humidity low. Basement labs with leaky pipes or sweating walls end up regretting it—chemicals degrade, labels smear, inventory gets costly or dangerous.
2,6-Dichloropyridine shouldn’t sit next to acids or strong oxidizers. Mixing these in storage makes cross-reactions far more likely, and with toxins like this you just can’t take that chance. I’ve seen careless stacking in cramped storage rooms, and it only needs one rushed hand to send powder flying onto the wrong shelf. Segregation and labeling never seem urgent until the day a spill reminds everyone why the rules exist.
Plastic, glass, or metal drums with tight seals work best. The manufacturers know their stuff here—repackaging into something “easier” almost always backfires. I once saw a batch diverted into old soda bottles for “convenience”; the results weren’t just unsafe, they were expensive after everything had to be cleaned up. Keep compounds in original containers with the label visible, and make sure staff actually read them. It’s common sense but surprisingly rare.
Disposal gets little attention until bins start to fill up. For 2,6-Dichloropyridine, tossing it in the regular trash brings serious legal and environmental risks. Specialized chemical waste streams—run by trained staff—are the only safe bet. Lab workers sometimes believe a detergent wash solves everything, but this stuff lingers in drains. It’s easy to forget about downstream impacts, but one slip can spoil local water—and with today’s scrutiny, word spreads quickly.
People in labs and storage rooms don’t gain good habits overnight. Regular talks about what’s at stake, plus simple, clear signage, make more difference than another round of dry compliance training. I’ve found that when staff see the rationale behind protocols—not just the “what” but the “why”—they start looking out for each other, not just themselves. That’s what real safety culture looks like, and that’s what prevents most problems with chemicals like 2,6-Dichloropyridine long before they start.
Factories and labs use 2,6-Dichloropyridine for a range of jobs, mostly as a building block for drugs, dyes, and pesticides. At first sight, the name alone throws up red flags for most people. Chlorine atoms bolted onto a pyridine ring might not sound as frightening as some of the more notorious chemicals, but that doesn’t mean it gets a free pass. Chemicals aren’t just scary on paper—they can be dangerous in reality, especially if the way people handle them doesn’t keep pace with what we already know.
Digging into its track record, information from producers and safety advisories point out a few basic facts. 2,6-Dichloropyridine is not going to jump out of a bottle and hurt someone by itself. Still, the substance can irritate skin, eyes, and lungs. The real trouble starts with consistent or careless exposure. Workers mixing or transferring the chemical risk breathing in dust or fumes, especially when ventilation lacks. I remember walking through a small formulation facility years ago—the air sometimes held a faint musty odor from solvents and handling flubbed bags of powders left some techs with red, itchy arms. Nobody likes sitting in a doctor’s office explaining how they got a chemical rash.
Folks in chemical plants soon learn that gloves and masks are not up for debate. No one wants chemical burns, and everyone wants their shifts to end without a trip to the emergency room. A review of 2,6-Dichloropyridine’s documentation shows that direct skin contact can cause nasty irritation; inhaling it can bring on coughing and respiratory pain. There are stories around the industry about workers who shrugged off those goggles and regretted it—the eye discomfort alone sticks in memory.
Things get more concerning with accidental spills or fires. This chlorinated compound releases hazardous gases, including hydrochloric acid and nitrogen oxides, if it burns. I’ve seen the aftermath of a chemical warehouse fire: the clean-up took weeks, and no one who went through it takes “fire protocol” for granted anymore. Safety professionals recommend storing it away from anything that might spark a blaze, and keeping water handy doesn’t hurt.
Long-term effects haven’t been studied as closely as with some bigger-name toxins like benzene. There is a gap that worries toxicologists, especially with more companies using this compound. Animal testing points toward risks beyond just skin irritation, but there’s still no evidence for cancer or major organ damage in people—at least not that’s been published in respected journals. That doesn’t mean all-clear, just that the research hasn’t caught up yet.
I’m a believer in respecting chemicals for what they are, not what marketing says. For 2,6-Dichloropyridine, that means sticking with good ventilation, sturdy gloves, and respecting the safety data. I’ve seen how skipping those basics goes after just a few months of working in a hurried environment. Regular safety training and sharing stories of real workplace mishaps help people remember why these procedures exist.
2,6-Dichloropyridine has its place in industry, but its risks deserve more voice than they sometimes get. Anyone handling it should have straight-talk on safe handling, plus updates whenever new research lands. Most chemical hazards sneak up on those who drop their guard. Respect plus solid facts—that’s what keeps workplaces safe, not blind faith in procedures or luck.
Walking through the maze of modern chemical research, I've noticed that some compounds keep popping up, even if their names rarely crack the headlines. 2,6-Dichloropyridine is one of them. I’ve spent enough time alongside chemists to see its name scribbled into lab notebooks, taped on bottles in storerooms, and threaded through patent reports. Most folks outside the field never realize how often this obscure-sounding chemical shapes products and processes they run into every day.
Medicinal chemistry continues to lean heavily on building blocks like 2,6-Dichloropyridine. It’s such a reliable starting point for preparing molecules aimed at disease-fighting drugs. Pyridine rings show up in compounds for everything from antibiotics to blood pressure pills. When you tack on chlorine atoms at the right places, that core suddenly snaps into new roles—sometimes serving as the backbone of an antiviral or helping craft molecules that control inflammation.In my time shadowing researchers on late nights, some of the most interesting discoveries grew out of “swapping” these halogens during synthesis. Yanking hydrogen out, plugging chlorine in—it can make all the difference between a dud and a blockbuster medicine. Chemists need these nuances. If the supply of intermediates like 2,6-Dichloropyridine dried up, the new-drug pipeline would sputter and slow, especially at the early testing and patenting stages.
Agriculture has its own quiet reliance on this compound. Out on the fringes of big seed companies, crop scientists look for ways to shield food supplies from weeds and bugs. Herbicides and pesticides that stick in the soil just long enough—without causing harm down the food chain—often trace some of their origins back to chlorinated pyridines. Large-scale production of such agents pulls directly from the chemistry built around 2,6-Dichloropyridine.I’ve seen the fine line these applications walk. Overdoing it can push risks up the chain, so careful, measured use carries major weight. Regulatory agencies eye chemical residues, prompting researchers to focus not just on yield, but also on environmental behavior. The challenge never floats far from the supply of raw materials.
Industrial research often demands new flavors of polymers and specialty products—heat-resistant coatings, flexible electronics, adhesives that don’t wilt in rough weather. At the foundation, you’ll spot small building blocks like 2,6-Dichloropyridine, which let chemists create new properties in the plastics and resins everyone takes for granted. Even as tech advances, industries need a stream of complex molecules to keep pace with new demands for strength, flexibility, and longevity.
It’s easy to overlook supply chain pressures, but sourcing chemicals like 2,6-Dichloropyridine can get tricky when demand shifts or regulations tighten. As someone who has seen disruptions ripple through labs and production lines, I know how much—down to the gram—matters. Balancing cost, safety, and sustainability has always been a grind. Watching companies search out greener production methods feels encouraging. Sometimes, that means finding cleaner catalysts; sometimes, it’s about shorter reaction steps or reclaiming waste streams that once got dumped.If there’s any lesson here, it’s that the solutions come best from collaborations—scientists, manufacturers, regulators, and even ordinary citizens pressing for cleaner products. The quest to rethink chemistry never stops, and every piece of innovation ripples out, touching everything from healthcare to food to technology at a fundamental level.