The timeline of 2-Chloro-5-trichloromethylpyridine ties closely to the evolution of modern agrochemical synthesis. Back in the 1960s, the chemical research community in Europe and North America started paying attention to the pyridine ring and its derivatives. Papers from that era pointed out how tweaks on the pyridine skeleton—like substituting chlorine or trichloromethyl groups—could lead to compounds with new and powerful biological activity. This interest eventually led to discovery projects in major crop protection firms, who saw the potential in these chemicals as starting points for new herbicides and pesticides. Over the decades, process chemists refined production methods, scaled up yields, and cleaned up the outputs, chasing efficiency and compliance with increasingly strict regulations. Conversations from that period in the chemical industry often revolved around patent races, environmental concerns, and delivering the right balance of efficacy and safety.
People often see 2-Chloro-5-trichloromethylpyridine at the foundation of several agricultural chemicals, especially preemergent herbicides. Its reputation comes not from being the final active ingredient, but as a key building block in the syntheses of several crop protection agents. In the field or at the lab bench, chemists value its reactivity, which opens the door to a wide array of functionalization routes. Quite a few listeners at agricultural symposia recognize the name because of its contributions to weed management products and also in specialty chemical labs, where its chemical behavior continues to spark fresh interest.
A person who’s handled this compound in person notices quickly its pale yellow powder or occasionally crystalline form. The compound resists water but goes into common organic solvents. Its stability stands up under mild laboratory conditions, and the high chlorine content provides both reactivity and some challenge when it’s time to clean up. The compound’s substantial molecular weight and somewhat pungent smell call for careful storage and a healthy respect at the bench. People who work with it know to check for possible vapors—small scale or pilot, those precautions always count.
Many producers offer 2-Chloro-5-trichloromethylpyridine with a purity in the upper ninety percent range, targeting applications where impurities cause big differences in downstream reactions. Each batch comes tracked by an identifying number, reference to origin, and a set of analytical readings guaranteeing content and structure. Reliable suppliers make a point to flag regulated components, hazard pictograms, and detailed instructions for handling. Regulations in the US, EU, and China all demand clear labeling about acute toxicity, storage, and disposal, keeping manufacturers on their toes with updates as policies shift.
Process chemists rely on a chlorination route with a trichloromethylating agent, often in the presence of catalysts or solvents tailored for selectivity. The reaction sequence starts from a pyridine base routed through chlorination, which often chooses phosphorus trichloride or similar as the halogen source. From there, the trichloromethyl group attaches with the help of specific reagents under temperature-controlled conditions, minimizing byproduct formation. Getting yield and selectivity right calls for constant monitoring, and even small procedural tweaks can mean the difference between a clean isolation and a messy separation job.
In graduate research discussions, this compound stands out for its activity at the ring-substituted positions. The electron-withdrawing effect of both the chlorine and trichloromethyl groups increase the ring’s susceptibility to nucleophilic substitution, letting chemists explore a whole series of modifications. In practice, 2-Chloro-5-trichloromethylpyridine carries forward its trichloromethyl group into ring-opening or further halogenation, sometimes generating crowded reaction schemes that intrigue both synthetic and process chemists. With so many possible transformations, it acts as a fork in the road for diverse product pipelines in pharma and agchem alike.
Ask most chemical suppliers, and they’ll rattle off synonyms like 5-Trichloromethyl-2-chloropyridine, or 2-Chloro-5-(trichloromethyl)pyridine. Older papers may use alternate numbering based on different naming conventions, a point that sometimes causes confusion in literature reviews. The variety of product names in global supply channels depends on branding by chemical distributors, but the CAS number remains the main identifier for purchasers committed to avoiding cross-contamination between structurally similar pyridine derivatives.
Working with 2-Chloro-5-trichloromethylpyridine involves a mindful safety culture. Lab workers and operators typically keep to gloves, goggles, and a ventilated bench as minimum precautions. Regulatory bodies like OSHA and REACH require that exposures sit far below defined thresholds, and anyone who’s experienced a spill can tell you clean-up gets complicated quickly because of its persistence and potential for inhalation risks. Mixing or scaling up production calls for automated systems, closed transfers, and regular training drills so everyone on-site understands protocol. On the ground, people treat disposal of waste streams as a community responsibility, working with authorized handlers to keep environmental exposures to a minimum.
2-Chloro-5-trichloromethylpyridine’s biggest mark lands in agriculture, especially for the protection of groundnuts, soybeans, and corn. It figures prominently in the synthesis of herbicides such as dinitraniline-style compounds. Major agrochemical companies include it in multi-step syntheses where fine control of position and reactivity drives the final product profile. Beyond fields, research teams harness its ring pattern for pyridine-based syntheses, building blocks for veterinary products, and specialty materials. More recently, niche uses have emerged for the derivatized forms in advanced organic electronics and polymer chemistry, though these remain smaller markets.
R&D circles keep busy testing the possibilities for 2-Chloro-5-trichloromethylpyridine. Researchers from public and private sectors evaluate how its modifications can boost selectivity for weed targets or reduce non-target organism effects. Projects underway probe greener routes to synthesis, aiming for less energy consumption and cleaner streams. At the university level, curiosity drives discovery, and student teams tackle questions about new reactivity, catalyst optimization, and safer alternatives to hazardous reagents used in its preparation. Academics and industry lab partners share results at conferences, pushing thinking past traditional uses toward the next generation of pyridine chemistry.
Toxicologists track 2-Chloro-5-trichloromethylpyridine carefully. Lab results flag moderate acute toxicity if ingested or inhaled. Repeated exposure raises questions about organ specificity in mammals, with most regulatory agencies classifying it as a substance demanding strict handling for both humans and ecosystems. Early studies flagged bioaccumulation risks, leading regulators to tighten oversight on effluent disposal and occupational safeguards. Medical professionals on chemical safety teams point out the necessity for quick decontamination after skin or eye contact, and updated toxicology screens influence company-level decisions about inclusion in final product lines. Ongoing animal studies and monitoring of real-world exposure set the stage for future refinements in safe handling policies.
Looking forward, 2-Chloro-5-trichloromethylpyridine’s place in agrochemicals seems set, but market pressure for safer and more sustainable solutions will push manufacturers to rethink its role. Synthetic chemists currently design greener routes that deliver the same intermediate with reduced hazardous inputs and lower carbon footprints. In parallel, the move toward bio-based pest control nudges researchers to use classic molecules like this in smarter, more selective formulas or biodegradable derivatives. Regulatory trends keep shifting toward limits on persistent or volatile chemicals, so production cycles may see pressure to tighten emissions, recover solvents, and invest in closed-loop systems. For now, 2-Chloro-5-trichloromethylpyridine stands as a mainstay of chemical synthesis, but its future will rest on the balance between performance, safety, and the growing environmental movement shaping today’s global chemical industry.
Some chemicals don’t pop up in daily conversation, but their shadow lies over more things than we expect. 2-Chloro-5-trichloromethylpyridine stands out as one of those substances with a bigger story than its polysyllabic name might suggest. Tucked into the often-invisible world of modern agriculture and pest management, this compound shapes how farmers and scientists handle stubborn weeds and threats to crops we all rely on.
Practically speaking, 2-Chloro-5-trichloromethylpyridine lands in the middle of a chemical chain leading to widely used herbicides. Take the example of triclopyr—a common name for those in farming or gardening. The chemistry starts with the synthesis of this pyridine derivative, building up to weed control agents that tackle everything from invasive vines to brush threatening pastures. Farmers face persistent pressure to keep fields healthy, and these chemicals give them real options where manual labor would fall short, especially on a scale needed to feed cities.
I grew up near farms that dealt with noxious weeds like poison ivy sprouting up between crops. I remember neighbors wrestling with bent backs and gloved hands, digging roots only to see them return days later. The arrival of selective herbicides changed the game, offering not only relief but also putting food security on a firmer footing. Ingredients like 2-Chloro-5-trichloromethylpyridine form the scientific muscle powering these tools.
No tool comes without trade-offs. Synthetic chemicals—including those that start with this pyridine derivative—raise real concerns around water contamination, toxicity to fish or insects, and persistence in the environment. Runoff isn’t a hypothetical risk. Plenty of studies track the movement of herbicidal residues from fields into waterways. Some products stay in soil for months or longer. This persistence keeps weeds down but also sparks debate among biologists and public health groups. Safety data exists to guide use, but the landscape keeps shifting as more is learned about what these compounds do outside the farm gate.
Balancing crop protection with ecological responsibility always brings friction. In the past decade, greater scrutiny has landed on how much of any chemical ends up where it shouldn’t. It brings up tough choices. Ban a compound outright, and yields can suffer; keep using it recklessly, and the wider world pays the price in lost pollinators or tainted groundwater. So, the job of folks producing, buying, and spraying chemicals built on 2-Chloro-5-trichloromethylpyridine requires a steady hand and respect for both farm economics and soil health.
Some solutions grow from the bottom up. Researchers keep testing less persistent alternatives and promote integrated weed management strategies. Instead of spraying field-wide, precision agriculture now lets some growers target problem patches, trimming down widespread exposure. Buffer zones near streams and better application timing further reduce environmental drift. Public pressure and stricter regulation push manufacturers to rethink how these chemicals are produced and handled from day one.
The story of 2-Chloro-5-trichloromethylpyridine speaks to a larger pattern in food production—progress tied to chemistry, with new lessons and new risks at every twist. Informed choices, regular review of evidence, and honest conversations about safety mean more than marketing claims or fear-driven bans. This compound, like many others, sits in the middle of that ongoing debate: how to feed a growing population without losing sight of the fields and rivers that make it possible.
If you have spent any time in a lab, you learn pretty quickly that chemicals don’t forgive slips or shortcuts. 2-Chloro-5-trichloromethylpyridine is no exception—this compound plays a role in a range of research and manufacturing settings, and its hazards make careful handling a non-negotiable. Years of sharing cramped bench space with sharp-eyed chemists taught me: you never regret safe habits.
2-Chloro-5-trichloromethylpyridine falls into a category of substances that can irritate the skin, eyes, and lungs. Just a quick open of a bottle can push a pungent odor through the workroom—clear evidence that this isn’t something to take lightly. Spills don’t just evaporate quietly; they leave behind risks that linger on surfaces and in the air.
You want the basics in place before even flipping open a container’s cap. I always reach for nitrile gloves, a solid lab coat, and chemical splash goggles. The compound vapors can sting the eyes and nose, so good ventilation means more than just cracking a window. Fume hoods are your best friend. At one lab, a colleague decided to skip the hood “just for a minute”—the next morning, our whole team walked into a space that needed hours of airing out before we could get to work.
There’s a temptation to ease up when you get busy, but I’ve seen quick hand washes fail to remove chemicals that sneak through thin gloves or up shirt sleeves. Chemical-resistant gloves and snug-fitting sleeves keep exposure down. Anyone with sensitive skin knows that redness or rashes can flare up even after contact you barely noticed at the time.
If you knock over a beaker, no one benefits from panicked flailing. Loose absorbent pads and proper chemical waste containers should sit nearby. I remember a day that a junior member of the team had a minor spill and started cleaning up with just paper towels. The chemical cut through in seconds—burning her skin and eating at the desktop. Now, we keep spill kits at arm’s reach, and we all know how to use them. Fast, smart cleanup stops problems from spreading.
We all know what happens to bottles left unlabeled or jammed among acids and bases—unexpected reactions or nasty ruptures. I’ve seen 2-Chloro-5-trichloromethylpyridine bottles sweat and leak in the wrong conditions. This chemical prefers cool, dry, and out-of-the-way cabinets. Only compatible chemicals belong anywhere near it.
No manual covers every scenario. Newcomers in my past labs gained hands-on practice with mock spills and equipment before running into real risks. Refresher sessions every few months helped us stay sharp about the right steps, especially since guidelines evolve with new safety data.
No one wants to be “that person” who puts the team at risk. We nudged each other when shortcuts crept in. Over time, safety culture stuck because everyone made it part of the daily routine—simple checklists, double-checking labels, and walking each other through cleanup drills.
Regulations and chemical safety data keep shifting as more studies come out on compounds like this one. Trustworthy sources—peer-reviewed journals, regulatory agencies—motivate healthier skepticism and new strategies on the job.
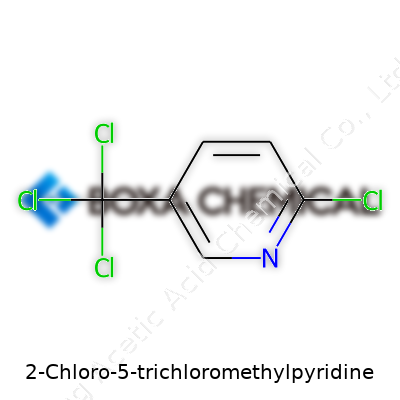
In the end, care and consistency keep you healthy—and the work rolling forward. Every time you handle something like 2-Chloro-5-trichloromethylpyridine, you’re not just protecting yourself, but everyone who works alongside you.2-Chloro-5-trichloromethylpyridine stands out because of how its atoms join, and the way those bonds shape its reactivity. You see, beneath its long name lies a ring of six carbon atoms, one swapped out for nitrogen. Folks who worked in a chemistry lab—or even took an organic chemistry class—might remember pyridine and its sharp, fishy smell. Chemists call pyridine an aromatic heterocycle: it’s got a ring with alternating double bonds, but nitrogen takes the spot of one carbon.
What sets this compound apart lies in what’s attached to this pyridine ring. At the second spot around the ring, a chlorine atom sits. At the fifth, you find a trichloromethyl group: that’s a carbon loaded up with three chlorines. So picture a hexagon (that’s the ring) with a nitrogen, a chlorine two spots from the nitrogen, and a three-chlorine cluster three spots further.
No chemist pays close attention to structure for the sake of tidy diagrams. The way atoms link up changes the behavior of a chemical. With 2-Chloro-5-trichloromethylpyridine, the extra chlorines toughen up the molecule. These atoms are electronegative, meaning they pull on the electrons shared in their bonds. They also shield the ring from attack by moisture, sunlight, or soil microbes.
This structure crops up in herbicides, and for a reason. Chlorinated rings hang around in the environment. They don’t break down easily, and that gives farmers a long-lasting tool against certain weeds. Regulators and scientists keep digging into the side effects, because resilient molecules can cause trouble if they linger where people or wildlife live.
Once I visited an agricultural science lab that measured tiny traces of chemicals in water, soil, and crops. The analysts worried about chlorinated aromatics sticking around too long and harming nearby plants that folks wanted to keep safe. They tested runoff from test plots sprayed with herbicides—plenty of which used cousins of this molecule—and found those chemicals in puddles days or weeks later.
Folks who deal with chemical management stress over breakdown rates. If fields stay residue-free, the job gets easier. If not, rules tighten up. In some regions, you can't use formulas containing persistent molecules like this unless it’s proven there’s no unacceptable risk. I’ve seen fields set aside after misuse or buildup—lost money for the farmer, headaches for regulators, and years of watching nature clear out the leftovers.
Chemistry gives us tools. It also lays out risks. Because the structure of 2-Chloro-5-trichloromethylpyridine helps it last, decision-makers need to set rules based on what science uncovers about where this compound ends up. Some labs are now testing options for faster breakdown—reformulating molecules, trying alternative groups in place of one chlorine, or adjusting the timing of application. More careful timing, using less, or switching to less stubborn structures can ease the load.
Learning about how bonds and atoms shape a chemical isn’t just for exam prep. It stays in the heart of decisions around food, water, and environmental safety. Solutions won’t only come from a new product, but from a shared effort between research, farming, and thoughtful policy.
Every time I walk past a storage shelf lined with chemical containers, safety and respect for the compounds inside come to mind. Chemicals like 2-Chloro-5-trichloromethylpyridine deserve particular attention due to their potent effects and risks. This pyridine derivative plays a role in manufacturing and research, but only disciplined care prevents trouble. Storing it without thought can spell headaches—both literal and regulatory. From leaks to fires or exposure, every mishap is a lesson on why rules around chemical storage exist in the first place.
Some products sit on the shelf and laugh off temperature swings. Pyridine derivatives rarely get that luxury. 2-Chloro-5-trichloromethylpyridine should be kept cool, away from heat sources, and far from direct sunlight. This isn't just a matter of preserving shelf life—it's about halting unwanted reactions that heat can spark. Excess warmth nudges volatility and can lead to pressure build-up in containers. In my lab days, stories spread about glass bottles cracking under summer sun because someone ignored this advice.
Moisture and this chemical never mix well. Water can prompt breakdown, starting reactions that alter the compound or create corrosive byproducts. Desiccators or low-humidity storage rooms fend off this threat. Even humidity creeping in through a carelessly sealed cap finds a way to do its damage over months. On more than one occasion, I've opened a supposedly safe bottle only to spot residue inside—clear evidence that moisture had gotten its way. Providing a dry and tightly sealed container offers better odds for long-term safety.
Many chemicals spark trouble not by their own design, but because of neighbors on the shelf. Strong acids, bases, or oxidizers can introduce unforeseen risks if housed nearby. Separation cuts down the risk of accidents if containers break or leak. I remember a mentor showing me shelves marked not just by inventory numbers, but by real risk relationships. Every line drawn kept incompatible neighbors apart for a reason. Segregation matters not out of tradition but out of lessons written in spilled chemical stains.
Glass usually wins out for chemicals like this, since plastics sometimes get eaten up by strong organic compounds. Tightly sealed lids are a must, blocking both air and water vapors. Labels need to be clear and up to date—life moves at its own pace and forgetting what's inside a dusty bottle only courts disaster. Poor labeling practices once cost me precious hours in the lab, sorting out which bottle held which chemical, and the memory still stings.
Regulators don’t draft their requirements out of thin air. Rules around 2-Chloro-5-trichloromethylpyridine exist because past mistakes created a blueprint for caution. Training and signage help, but real safety relies on building habits—closing caps right away, checking for leaks, making sure that storage areas get regular inspection. Following these steps isn’t red tape; it’s what keeps people safe and science moving forward without avoidable setbacks.
Safe storage gives back more than lost hours or wasted product—it protects people and the environment from unnecessary harm. Even outside a research setting, chemical stewardship reflects a deeper respect for science as a human endeavor. Paying attention to storage conditions for 2-Chloro-5-trichloromethylpyridine doesn’t take extraordinary effort, just steady routines and a mindset that refuses to cut corners. That’s the foundation of real expertise and trust in any lab or industrial space.
Most people rarely give a thought to the chemical building blocks inside the pesticides and herbicides designed to keep crops free of weeds. One of those blocks—2-Chloro-5-trichloromethylpyridine—serves as a key raw material in the creation of many commercial weedkillers, especially those labeled for use on cereal crops and turf. Modern farming leans hard on these substances to keep harvests viable, but that relationship comes with environmental baggage.
The story doesn’t stay neatly contained in the fields. After application, chemicals like 2-Chloro-5-trichloromethylpyridine often stick around in the soil far longer than most shoppers or even many local farmers realize. Studies and real-world reports show that some of the related compounds persist in the ground for months—or even years—before breaking down. That’s not a small detail when you consider what sticks around in topsoil also finds its way into our water, wild plants, and even dust in the wind.
This compound and its downstream products have the reputation of making life tough for certain kinds of soil-dwelling life. Earthworms, beneficial microbes, and insects all get caught up in the fallout when soil chemistry changes sharply. In those situations, soil loses a chunk of its ability to recycle nutrients, support roots, and resist floods or erosion. Over time, fields risk becoming less productive or more reliant on artificial supplements just to maintain crop yield.
After heavy rainfall, surface water runoff can carry both active and broken-down chemical traces into streams and rivers. Some researchers record worrying results in aquatic life, such as inhibited growth rates or disrupted reproductive cycles in fish and amphibians. Even small traces of this class of chemicals stack up over time, raising alarms about broader ecosystem effects that stretch beyond one harvest.
My own conversations with farm advisors across different regions usually touch on stories about seeing frogs become less common or anxious questions about water quality near old spray zones. Nobody likes to see downstream effects pile up after a treatment that was meant to help crops thrive.
Environmental agencies keep track of residues for a reason. In Europe and parts of North America, strict limits set which chemicals can be sprayed and in what quantity. Some governments have already placed heavy restrictions on certain pesticides containing 2-Chloro-5-trichloromethylpyridine after linking them to sluggish breakdown in the soil and risk to ground water. If a compound lands on watch lists, it’s usually for good cause. Decades of monitoring establish that some breakdown products don’t just vanish. Instead, they linger in water supplies and can show up in food chains.
Solutions don’t just land with one ban. Innovations in precision agriculture help farmers use chemicals more responsibly, applying the bare minimum. Researchers work on alternative methods—planting cover crops, rotating species, or experimenting with biological pest control techniques. These options take effort and investment, which means support from governments and communities holds weight.
Clean soil and safe water benefit everyone, not just people who make their living from the land. As the evidence stacks up around the baggage chemicals like 2-Chloro-5-trichloromethylpyridine bring, society faces a choice about how much risk to accept. Exploring and investing in smarter, safer crop management is not just possible—it’s necessary if we want tomorrow’s harvests to feed us without long-term costs nobody signed up for.