Stepping back through the pages of industrial chemistry, 2-Isopropyl-4-methyl-6-pyrimidinone hasn’t made front-page headlines, but its history mirrors the discipline’s steady advance since the mid-20th century. Pyrimidinone scaffolds drew attention as chemists chased after molecules with new pharmaceutical and agrochemical potential. During the wave of research into heterocyclic compounds in the 1960s and 1970s, this particular derivative surfaced in patent filings and scattered journal notes, quietly laying the groundwork for applications that would blossom years later. My mentors in research often spoke of sifting through shelves filled with odd, latent compounds, and 2-Isopropyl-4-methyl-6-pyrimidinone sat among those, waiting for the right questions to be asked.
What pulls me to this compound is its adaptability. 2-Isopropyl-4-methyl-6-pyrimidinone offers a stable core that resists rapid breakdown, making it valuable for chemists striving for controlled reactivity. The ring system carries a unique balance—neither too reactive nor inert—allowing for thoughtful design in synthesis. This isn’t one of those blockbuster molecules touting miracle properties, but rather a versatile workpiece: a building block appreciated among researchers who recognize that breakthroughs often start with sturdy, reliable chemistry.
Anyone who has handled 2-Isopropyl-4-methyl-6-pyrimidinone in the lab remembers its pale crystalline appearance. It’s not volatile, so it doesn’t fill the air with fumes or odors, and its solid state makes weighing and measuring straightforward. Water won’t do much to dissolve it, but organic solvents take it in willingly enough. The molecule’s fused ring system brings a chemical resilience, allowing it to stand up to moderate heat and resist decomposition under standard bench conditions. Its melting point falls in the moderate range, right where you want it for material handling without fuss or risk.
Clarity on technical specs matters for safety and for reproducible results. Most suppliers stick to purity ranges above 98 percent, since impurities muck up downstream reactions or analyses. Package labeling speaks plainly: chemical name, structural formula, hazard warnings where needed, batch numbers for traceability, recommended storage conditions. That attention to labeling isn’t just bureaucracy—it reflects the real-world need to avoid dangerous mix-ups, as any busy synthetic chemist will attest. Over years spent in shared lab spaces, I’ve seen what slips through when labeling standards drop, and it’s never worth the risk.
Synthesis often begins with pyrimidine precursors modified through alkylation and oxidation, a path well-mapped by practitioners of heterocyclic chemistry. Common strategies draw on base-catalyzed reactions, making use of relatively simple reagents to introduce the isopropyl and methyl groups at precise positions. Once you’ve run enough experimental batches, the logic behind the steps clicks: each condition tailored to coax this molecule into its final form and keep yield up, byproduct down. I’ve sat with friends in grad school troubleshooting temperature swings or trying to optimize those exact steps in the process—the preparative method turns into a kind of ritual, repeated until the product reliably appears.
The value of 2-Isopropyl-4-methyl-6-pyrimidinone shines through most brightly once you start coupling it with other building blocks. That stable pyrimidinone ring resists harsh acids and bases, so it weathers multistep syntheses. Introduce functional groups at available positions without risking collapse of the structure. Over years, research teams have tried a range of substitutions—halogenation, amination, and acylation among them—to craft customized derivatives meant for deeper study or tailored applications. My own experience with such chemistry taught me that minor tweaks to the ring’s decorations open new doors in medicinal and agricultural chemistry, often in unexpected ways.
The chemical goes by a mouthful of identifiers: one might encounter phrases like “isopropyl-methyl pyrimidinone” on research orders or regulatory filings, or various abbreviation games on data sheets. CAS numbers usually narrow it down fast for inventory tracking or regulatory compliance. With so many names floating around, cross-checking synonyms in databases has saved more than one project from redundant purchases or paperwork headaches. In conversation, most chemists default to the shorthand—they know a layered name signals more than just a molecular formula; it reflects family ties within the pyrimidinone world.
Nothing stalls a project faster than a safety incident. This is a solid, but gloves and goggles still come out by habit. If dust accumulates during weighing or transfers, those tiny airborne particles pose inhalation risks. Flammable solvents used alongside it mean static or open flames get real attention. Waste handling routines matter: disposal streams sorted, logs kept. Colleagues and I have trained new lab members on real-world dangers beyond the MSDS, stories of spills or forgotten gloves sticking as lessons that never fade. Standard operating procedures built over years by cautious hands have become second nature, and there’s no shortcut around them.
Most current research with 2-Isopropyl-4-methyl-6-pyrimidinone turns to pharmaceuticals and agrochemicals. Its ring structure mimics bioactive scaffolds, opening possibilities for drug development—especially for anti-infectives and enzyme inhibitors. I have seen this compound used as an intermediate in new herbicide projects, where targeted modifications extend its usefulness into crop protection. Colleagues in the materials science side have dabbled with it as a precursor for specialty coatings, though the scene there is still developing. Its adaptability rewards creativity, so fresh applications often stem from researchers willing to explore less-trod paths rather than sticking to published lit.
In the world of R&D, exploration means pushing beyond the known. Groups investigating new therapies trawl through libraries of heterocyclic compounds, pitting 2-Isopropyl-4-methyl-6-pyrimidinone against targets both clinical and agricultural. Years in corporate and academic labs have shown me that the “curse of abundance” can bog down projects—so many candidates, so little time. Still, this molecule’s track record in fragment-based drug discovery means resourceful teams keep circling back to it. It draws in both painstaking tinkerers and broad-minded modelers seeking a reliable scaffold within a surge of possibilities.
Experience has taught many researchers to respect the blind spots in toxicology. The literature on 2-Isopropyl-4-methyl-6-pyrimidinone’s toxicity continues to grow, but gaps remain. Laboratory test data suggest moderate toxicity, with particular care needed when scaling reactions. This isn’t a compound to treat lightly—the risks stack up in high-dose animal studies and cell assays. I recall mentors coaching teams to handle everything as if unproven hazardous, an approach that slowed no one but protected everyone. For now, routine exposure limits stay cautious. Long-term environmental impact and chronic exposure questions encourage even greater restraint in both experimentation and commercial rollout.
The future for 2-Isopropyl-4-methyl-6-pyrimidinone sits at the intersection of cautious optimism and uncharted territory. Demand grows for chemical cores that bridge cost, utility, and manageable risk—exactly where this molecule fits. I see real potential as research tools diversify and custom syntheses expand, especially as AI-driven screening and high-throughput biology unlock new uses for subtle ring modifications. Big gains could come from green chemistry methods that reduce byproducts and environmental burden, though such approaches demand time and hard-won insight. The discipline thrives on compounds with room for reinvention, and this pyrimidinone has the right bones for a new generation of problem-solvers looking for more than instant, dramatic results.
Stroll into a discussion with researchers focused on agriculture or organic synthesis, and 2-Isopropyl-4-methyl-6-pyrimidinone pops up sooner or later. Most people outside these circles have never heard of it, though proof of its impact sits in crops, greenhouses, and even pharmaceutical labs. This compound doesn’t grab headlines, but it’s held in high regard for a handful of reasons, especially in crop protection and new drug development.
For anyone who’s walked a field at dawn worried about wilting leaves, the ever-present threat from weeds, insects, and disease remains personal. Farmers rely on more than luck and tradition. Some crop protection tools bank on chemistry that starts with unique building blocks, and 2-Isopropyl-4-methyl-6-pyrimidinone wields plenty of value here.
It acts as a vital intermediate in synthesizing several herbicides, especially those tuned for selective action so crops grow while weeds wither. Pyrimidinone derivatives hang at the core of modern weed control that’s effective enough for major crops—think rice and wheat—yet designed to spare non-target plants. These molecules help tackle yield loss, keeping more food on tables with less land.
The U.S. Environmental Protection Agency keeps a close watch on ingredients feeding into herbicide processes, with ongoing studies dedicated not only to their immediate effectiveness but also to possible long-term effects. There’s no room for shortcuts with chemistry that winds up on our plates.
Medical science hasn’t ignored this compound either. Researchers studying antiviral and anticancer drugs see potential in the pyrimidinone core structure—it fits naturally into the chemistry already powering several life-changing medicines. Every week, scientific journals fill with new patent filings, and this molecule sneaks into the claims for a reason. Its framework can be tweaked easily, helping medicinal chemists switch up side chains and optimize drug candidates for real-world problems.
It’s no cure-all by itself, but as a building block, it can lead to breakthroughs that wouldn’t exist otherwise. Take the fight against emerging diseases: once a promising lead appears, labs scramble to create close relatives by shifting functional groups around the core. 2-Isopropyl-4-methyl-6-pyrimidinone plays a role in this by offering a foundation that’s ready to adapt.
Chemistry with such power comes with baggage. Producers need to take safety and environmental risks seriously—not only for those who make the stuff but for nearby groundwater and wildlife. Strict guidelines control how it gets handled and discarded, while companies seek out greener reaction pathways and less toxic solvents for its production. The push for sustainability shows up in these corners too: think enzyme-driven synthesis instead of energy-hungry classic methods.
My years spent walking alongside growers and watching pharmaceutical teams puzzle over molecular design put a finer point on things. Progress demands more than clever molecules—it asks for transparency in research, accountability from manufacturers, and smarter stewardship of our soil and water. Getting these factors to line up pushes everyone to do a little better each season.
As we keep our focus on feeding more people with less waste and offering fresh hope to patients, these under-the-radar chemical tools gain importance. The real challenge lies in using them wisely—balancing their possibilities with the needs of our communities and planet.
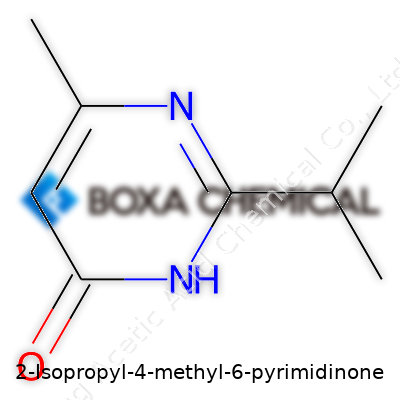
There’s something fascinating about seeing how rearranging a few atoms on a ring brings about a brand new molecule. 2-Isopropyl-4-methyl-6-pyrimidinone stands as proof. A mouthful to say, but fairly simple in structure for anyone used to staring down organic compounds. The core of the molecule is a pyrimidinone ring—a six-membered aromatic structure with two nitrogen atoms and a carbonyl oxygen. If you love patterns, this one’s pretty satisfying. The nitrogens park themselves at position one and three; the carbonyl group sits at position six. Toss in an isopropyl group at the second spot and a methyl group at the fourth, and you land right on 2-Isopropyl-4-methyl-6-pyrimidinone.
Chemical structure and substitution can look trivial—so you moved a few atoms around. But these tweaks change how a molecule behaves. Different groups at positions two and four amp up the character, modifying its reactivity, melting point, and what it does once it lands in the lab or the environment. For folks who spent years fidgeting with molecular models or watching NMR spectra, these details shine. The isopropyl group, with its Y-shaped formation, bulks up that second ring site. This sort of group likes to add stability or soften harsh reactivity, depending on its neighbors. Over on the fourth carbon, the methyl tucks in, offering up just a little hydrophobicity without crowding the rest of the structure.
I remember my first encounter with pyrimidinone derivatives in a dusty graduate lab. We puzzled out why one fluorinated molecule stuck to enzyme pockets better than another. Tiny substitutions means a huge difference for properties like solubility, what kind of reactions are possible, or whether a certain enzyme latches on. Structure means destiny—especially when you’re working with molecules this precise.
It’s not just an academic exercise, either. Changes to a chemical backbone affect safety, biological effects, and what industries do with it. Pyrimidinone rings form the core of several drugs and crop protection products. Even a small modifier like an isopropyl or methyl can tweak toxicity, how fast the molecule breaks down, or where it tends to accumulate. Safety regulators dig into these details, using rigorous studies and validated measurements, such as logP values or metabolism rates. Getting a handle on chemical structure makes it possible to predict risk and tailor appropriate safety measures.
One big issue tied to compounds like this is potential persistence in water and soil. Some substituted pyrimidinones resist microbial breakdown. Scientists are looking at new predictive models, hoping to gauge environmental impact before hundreds of tons reach the market. Structural awareness gives industry and regulators a conversation starter for better stewardship. Green chemistry offers alternatives where designers swap in groups that hold onto performance but degrade faster. With every new substitution, there’s an open invite to balance innovation with responsibility. Structure doesn’t just belong on chalkboards; it holds real-world consequences everyone shares.
Over the years working in research labs and talking shop with chemical handlers, I've learned that proper storage isn't just about following a checklist. With a compound like 2-Isopropyl-4-methyl-6-pyrimidinone, risk creeps in from basic mistakes—people stash bottles on open shelves, forget to check seals, or turn a blind eye to temperature swings. Such habits led to lost product, ruined experiments, and real safety threats.
Keeping this chemical away from moisture and sunlight is more than a routine; it protects both the integrity of the material and the people in the lab. Even minor exposure to dampness or heat can degrade many organic solids, and pyrimidinone derivatives aren’t known for resilience against careless treatment. In my own work, I’ve seen freshly purchased reagents go to waste because lids weren’t tightened or someone moved flammable materials next to oxidizers just to clear space.
I prefer handling this sort of compound in a cool, dry spot—usually a cabinet with solid doors, far from direct sunlight and potential heat sources. Dry boxes, fitted with desiccants, really pay off in humid climates. I always label my containers with the date and type of chemical, so colleagues don’t mistake the contents and nobody ends up opening an aging jar thinking it's sugar.
Good practice teaches the importance of secure closure. One time, a slightly loose cap let ambient moisture sneak in overnight, producing clumps instead of powder. That’s money out the window and wasted time. Hand-tightening works, but a quick double-check truly matters.
Temperature control often gets ignored, but temperature shifts can quietly break down chemicals, especially organics like this one. Stashing inventory near hot water pipes risks slow, undetected loss of quality. I’ve worked at facilities where basic thermometers next to storage areas helped spot bad habits. Room temperature usually works, but cold storage sometimes extends the product’s shelf life. It’s worth checking recent data, since published stability studies offer clues about the best ranges.
Handling nitrile gloves and goggles shouldn’t feel optional with chemicals like these, even if the MSDS sheet looks benign. In one instance, a simple glove swap saved my skin when I caught a container leaking. The minute I discovered a small spill, a grab for paper towels would have spread it and made cleanup miserable, possibly exposing my skin. Using the right absorbent pads and never touching a spill barehanded shows common sense at work.
Some labs try to cut corners on safety equipment or forgo spill kits, thinking nothing bad will happen “this time.” My view: spending a few pennies on extra gloves, fresh goggles, and proper storage beats facing an expensive cleanup or, worse, a health scare.
Training new colleagues often means repeating advice: Don’t overcrowd shelves, rotate inventory, and check everything twice. Experienced staff sometimes grow complacent, but the discipline to follow protocols creates a safer, more reliable workplace, which keeps projects on track.
Stories about chemical mishaps circle through the research community for good reason. Routines protect both health and investments. The extra three minutes spent labeling, sealing, and organizing pays off, project after project.
In any lab or industrial setting, people expect clear information about the chemicals around them. 2-Isopropyl-4-methyl-6-pyrimidinone doesn’t show up in everyday headlines, but every lesser-known compound deserves a spotlight on safety. Skipping that helps no one. Chemicals just as obscure have tripped up whole industries before, from early aniline dyes in the 19th century to more modern solvents and reagents.
I keep bumping into the same roadblock with this compound: a real lack of robust published data. Standard chemical safety databases (like PubChem, the National Library of Medicine’s TOXNET archives, and ECHA) tend not to list extensive hazard statements or toxicity outcomes for it. That says a lot. A compound in industrial or pharmaceutical chemistry that flies under the regulatory radar should set off a few alarms—mainly because gaps in data carry hidden risks, even if no red flags are flying yet.
Anybody who’s ever worked with untested chemicals knows the drill: assume the worst, not because of paranoia, but out of respect for the unknown. Pyrimidinones as a class don’t appear in the list of notorious toxins, but small molecular changes often flip the safety script. 2-Isopropyl and 4-methyl substitutions change bioactivity and, potentially, environmental behavior. With no direct studies on its acute toxicity, reproductive risks, or persistence in soil and water, there’s no credible way to declare it safe just because nothing bad has popped up.
History keeps giving us examples. Think about dichloromethane: lax early assumptions gave way to strong warnings after real-world exposures, decades later. 2-Isopropyl-4-methyl-6-pyrimidinone hasn’t carried that kind of baggage—not yet. That doesn’t mean workers or researchers get a free pass to skip gloves, ignore masks, or push leftovers down the sink. Even experienced chemists have overlooked hazards with “novel” compounds and paid a steep price. Responsible handling comes first—good ventilation, eye protection, and skin coverage are non-negotiable even when the data pool looks shallow.
Some companies don’t want to pay for extra testing until laws make them. That leaves workers and the environment holding the bag. Broader, transparent testing—acute toxicity in mice, irritation studies, ecological assays—should happen long before scale-up or mainstream adoption. It doesn’t take a disaster to start digging for answers. As more compounds enter the market, especially in pharmaceuticals and specialty chemicals, people deserve public and peer-reviewed safety data rather than promises and marketing optimism.
In my own experience, skepticism saves lives. If you haven’t found strong toxicity evidence one way or another, treat new chemicals as potentially harmful until proven otherwise. That means leaning on chemical fume hoods, keeping spill kits topped up, and refusing to shortcuts around disposal instructions. Regulators could encourage this mindset by requiring open reporting and supporting cross-border chemical tox databases.
No news often means nobody looked hard enough. Until independent toxicology work fills in the blanks, giving 2-Isopropyl-4-methyl-6-pyrimidinone extra respect in the lab isn’t alarmist—it’s just smart. Safety grows from asking tough questions, demanding solid evidence, and putting people’s health ahead of convenience every single time.
Staring at a chemical catalog, anyone working in a lab knows the chemistry section can easily feel like a maze. You see names like 2-Isopropyl-4-methyl-6-pyrimidinone, glance at the available grades, and find numbers like “98%” or, if lucky, “99%.” While these numbers might seem like bean-counting, there’s real meaning in that small difference, especially for researchers who want to make sure every experiment counts—or for manufacturers watching out for the tiniest impurities in each batch.
Chemists measure purity because even a hint of the wrong molecule can send a synthesis sideways. In pharmaceutical work, an unwanted contaminant, no matter how slight, can turn a promising project into wasted weeks. Many scientists I’ve worked with keep strict logs on every bottle, making sure that what’s inside matches the label. That’s not just fussiness; it’s about reducing the number of unknowns. With molecules like 2-Isopropyl-4-methyl-6-pyrimidinone, which can act as building blocks for more complex organic compounds, that reliability creates a sense of trust—a small but vital safeguard.
Looking at what’s usually available, chemical suppliers tend to offer 2-Isopropyl-4-methyl-6-pyrimidinone in purity levels upwards of 98%. These lots typically arrive in solid crystalline form. High-purity batches improve reproducibility in academic labs and scale-up environments. Sometimes, the vendor provides a certificate of analysis detailing exactly how they measured that number: perhaps thin-layer chromatography, NMR, or HPLC for purity checks. That transparency means more certainty for decision-makers who have to justify their material choices to regulatory teams, peer reviewers, and the occasional skeptical supervisor.
Ingredients like this rarely come alone. Specifications sometimes list acceptable ranges for physical characteristics—think melting point or color appearance. Some offer insight into trace elements. In an age when so many discoveries depend on tight tolerances, even a minor detail like residual solvents demands attention. Unchecked, that could spell trouble in sensitive catalyst studies or pharmacological screens.
Online, scientists swap horror stories about unexpected impurities. A friend once described a spectral analysis gone bad because the reagent had absorbed moisture due to a poorly sealed bottle. Simple attention to specification at the start can sidestep these headaches. Those who’ve spent enough time troubleshooting irreproducible results know that it’s often the “invisible” chemicals that create the loudest problems.
To see improvement, suppliers could publish more detailed batch records—think exact impurity profiles—not just broad-strokes purity percentages. Wider sharing of analytical data would help buyers sort out which material fits their purpose, and maybe even push manufacturers to tighten their production processes. For demanding projects, chemists might turn to secondary purification steps, or buy extra analytical equipment to double-check in-house. It’s a time and money tradeoff, but the peace of mind outweighs lost hours.
Purity and clear specification give projects the best shot at success. They also offer a silent guarantee that backs up every experiment or production run. As more research hinges on ever-finer details, those numbers mean more than just ink on a label—they shape results behind the scenes every day.