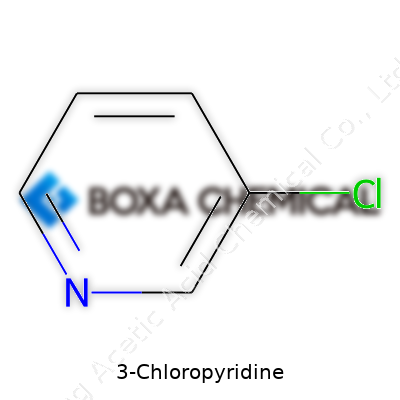
My first encounter with 3-Chloropyridine came late one evening in a university lab. The faint scent hanging in the air told its own history—this compound has been part of chemical workbenches for decades. Back in the golden years of organic synthesis, the race for functional pyridines drew researchers deeper into halogenated aromatics. By the mid-twentieth century, chemists had mapped out chlorination of pyridine rings, uncovering a family of derivatives with huge practical potential. The position of chlorine on the heterocycle, particularly at the third carbon, paid off with versatile synthetic handles. Fast-forward to recent years, screens flicker with research articles showing 3-Chloropyridine underpinning new agrochemicals, pharmaceuticals, and advanced materials. Seeing the compound’s journey from chemical curiosity to essential intermediate really grounds its importance in innovation—highlighting not just raw science, but the steady hand of progress steering new ideas into practical solutions.
3-Chloropyridine, as the name suggests, comes from the parent pyridine structure, swapping one hydrogen atom for a chlorine at the third carbon position. What stands out to me about this compound isn’t just its molecular profile—it’s what that single switch enables. Its presence in laboratory shelves speaks to a broader story of necessity: a chemical that opens the door for building massive, more complex molecules. Compared to other chloropyridines, this isomer gives good reactivity while holding down manageable toxicity, which counts for a lot if you’ve ever worried about handling more hazardous halogenated compounds. Its liquid state at room temperature also keeps lab work nimble, never getting in the way of pipettes and glassware. The versatility sits not just in its own reactions, but in what it lets chemists create after a few careful steps.
In practice, 3-Chloropyridine pours as a colorless or slightly yellowish liquid with a recognizable, sharp odor. The compound packs a molecular weight just shy of 130 grams per mole, with a boiling point around 170°C—meaning it holds together well under average heating, without jumping too easily to vapor. The density falls close to 1.2 g/cm³, so in most lab setups, it’s comparable to water, though you never want to take shortcuts with mixing or disposal. With a moderate solubility in water and much higher solubility in organic solvents (like acetone and ethanol), its behavior matches most chemists’ expectations for a small aromatic ring. A key feature: the chlorine atom at the third position tunes the electron density, dialing up reactivity at adjacent sites while holding resistance at others. This selectivity gives synthetic chemists opportunities for highly targeted transformations, though it demands proper controls for safe operation.
You’ll rarely find two bottles labeled the same way, but what matters remains: purity, batch, and hazard symbols. A reliable supplier provides 3-Chloropyridine at purities greater than 98% for most synthetic work. Labels flash pictograms for flammability and toxicity, as they should, and every lab vet pays respect to the safety data sheet before uncapping the bottle. Out in industry, technical documents dive into residue tests, impurity profiles, and shelf life—since a stray contaminant can derail downstream reactions fast. Regulatory lines draw bright boundaries too. Control measures frame this compound under hazardous substance rules, keeping workers aware of inhalation and skin contact risks.
Chemists have taken several routes to make 3-Chloropyridine. Older laboratories might chlorinate pyridine directly with chlorine gas or phosphorus pentachloride, though this scatters chlorines across multiple positions. More selective methods came along with catalytic approaches, starting from precursors like 3-pyridinol, running through chlorination with agents like thionyl chloride or phosphorus oxychloride. I remember supervisors shaking their heads at inefficiencies—yields ticked up only with tighter controls and better catalysts. Today, large-scale producers squeeze out cost savings and greener footprints using optimized flow processes and minimized byproducts. The challenge: juggling selectivity, waste management, and energy. Those synthetic achievements remind us how far process chemistry has come from brute-force days to today’s fine-tuned balance between output and environmental stewardship.
What draws many to 3-Chloropyridine isn’t just its existence, but the doors it opens. The molecule’s chlorine actively participates in nucleophilic aromatic substitution, allowing direct replacement by amines, alkoxides, or thiols under the right conditions. I’ve watched teams swap in complex side chains in just a matter of hours, saving cycles of protecting and deprotecting. More adventurous routes spark carbon-carbon couplings through Suzuki or Buchwald-Hartwig reactions, plugging in new cores for experimental drugs or agrochemicals. Reduction can strip the chlorine, returning to pure pyridine, but that feels almost a waste when the substitution drives so much forward chemistry. As research presses on, modifications like oxidation, reduction, and even lithiation show just how central this building block is in organic transformations.
You’ll meet this compound under several labels, including 3-chloropyridine, m-chloropyridine, and pyridine-3-yl chloride. These synonyms live side by side in literature and databases, sometimes tripping up new researchers in search engines. In industry, the simplicity of the IUPAC name sticks, though catalogues sometimes introduce translations or company-specific codes. The lesson fits any chemist: double-check synonyms during literature reviews and ordering, or risk wasting days to an unexpected isomer.
No glossing over this point—3-Chloropyridine commands respect for safe handling. The liquid’s volatility means a spill fills air with sharp, unpleasant fumes, quickly irritating eyes, lungs, and skin. On more than one occasion, I’ve seen experienced coworkers scramble for ventilation after a mishap. Regulatory bodies list this compound as hazardous: the safety data sheets warn about acute toxicity after inhalation and ingestion, and report irritation on contact. Gloves and goggles aren’t suggestions; they’re non-negotiable. Fume hoods and proper waste protocols make the difference between business as usual and a medical incident. Good labs keep antidotes, eye-wash stations, and spill kits ready—routine, not afterthoughts.
Pharmaceutical researchers lean heavily on 3-Chloropyridine as a backbone for medicinal chemistry. Its role as a synthon lets chemists swap in critical functional groups, creating molecules with tailored biological profiles. Major drug discovery projects often run many analogues off the same chloropyridine template to probe improved activity or metabolisms. In agriculture, the compound dials up pest-resistant chemistries—serving as a launchpad for new herbicides and fungicides that push crop yields higher. Advanced materials specialists draw on this building block for liquid crystals and electronic components, its nitrogen core bringing stability and polarizability. As needs evolve, so do the applications, knitting this compound into an ever-wider network of real-world problem solving.
Academic and industrial labs regularly push boundaries with 3-Chloropyridine. The latest literature tracks tweaks in selective chlorination, more sustainable catalysts, and low-waste processes. Collaboration between scale-up engineers and researchers trims bottlenecks, cuts emissions, and drives down costs. Creative surface chemists adapt the compound for sensor design, harnessing unique electron movements in sensitive arrays. Medicinal chemists chase new substitution strategies to build small molecule libraries for screening. Time and again, the utility of 3-Chloropyridine demands smarter approaches—never standing still while fields like green chemistry set new benchmarks for safety, efficiency, and lower environmental impact.
Toxicologists continue testing this molecule for acute and chronic hazards. Animal models illuminate risks after inhalation, ingestion, and dermal contact, helping set occupational exposure limits. Studies reveal irritation at modest concentrations, and higher doses have neurotoxic and hepatic effects. No lab I’ve worked in takes those warnings lightly. Waste management policies, air monitoring, and worker training shape a culture of caution. Modern research now digs into environmental fate: persistence in soils, mobility in water, breakdown by bacteria or UV. As regulations tighten, understanding these properties means improved stewardship—a duty owed to colleagues and surrounding communities.
Looking down the road, 3-Chloropyridine holds promise in fields not yet mapped. As sustainable chemistry progresses, efforts focus on making its preparation greener and its applications safer. The shift toward bio-based feedstocks and novel catalysts will likely redefine its industrial footprint, shrinking emissions while raising output. I see pharmaceutical development reaching for smarter modifications to the pyridine scaffold—amplifying therapeutic effects without sacrificing safety. Researchers tackling environmental issues will want new breakdown pathways and recycling options, making sure advances never leave toxic legacies. As someone familiar with both benchwork and broader impacts, I know every step forward with this compound will reflect not just technical progress, but a shared responsibility to use powerfully effective chemistry with care and respect for its consequences.
If you peek inside a laboratory focused on pharmaceuticals or specialty chemicals, you’re going to find bottles and drums labeled with names that don’t roll off the tongue. 3-Chloropyridine is one of those chemicals that probably sounds like it belongs in a dusty shelf, but the reality is quite different. Even though many people never hear about it outside technical circles, this compound matters far beyond its name.
3-Chloropyridine shows up as an intermediate—basically a stepping stone—in the manufacturing of many other chemicals. Drug development relies on it. Agricultural chemistry does too. Pyridine rings, like the core in 3-Chloropyridine, are a workhorse in synthetic pathways—think of them as Lego blocks for building new molecules.
A key fact about 3-Chloropyridine: chemists use it to make drugs that fight diseases in humans and animals. You’ll find derivatives of 3-Chloropyridine showing up as building blocks for antihistamines, antivirals, and even cancer treatments. My own experience working near pharmaceutical researchers showed how often projects start by transforming a molecule like this into something that ends up in a medicine cabinet. It saves time, cuts down on cost, and offers a predictable approach. This helps companies bring life-changing treatments to market faster.
Farmers also benefit from chemicals that start out as something like 3-Chloropyridine. The agrochemical industry uses it to build safe and potent pesticides and herbicides. With growing pressures on food supply, and as climate patterns keep changing, the need for stronger protection against crop disease and pests grows too. These chemical building blocks help address the challenge above and below the surface.
Dealing with 3-Chloropyridine is not simple. In the lab, researchers handle it with gloves, proper ventilation, and careful storage. In industrial settings, there’s a constant focus on minimizing worker exposure and preventing spills. Reports from regulatory agencies show that, although this chemical is useful, it carries hazards for workers and the environment if mishandled.
Companies which handle 3-Chloropyridine track emissions and waste closely. Neglecting those steps leads to real risks. I remember a conversation with a chemical engineer who described investing in improved scrubber systems and regular spill response drills. It’s not about checking regulatory boxes, but making sure health and the local environment don’t pay the price.
The story around 3-Chloropyridine pushes for more transparency in the chemical supply chain. Increasingly, stakeholders want to know—where do ingredients come from, how are they handled, and what risks crop up along the way? Maintaining public trust demands more than just technical compliance. It involves open dialogue and honest reporting.
Safer chemistry matters too. Scientists worldwide keep searching for alternative routes that cut down hazardous byproducts or use greener starting materials. Public funding and consumer demand for safer products accelerate that push. From my time observing R&D teams, I saw the most meaningful breakthroughs when researchers worked together rather than siloed off in separate labs or companies.
3-Chloropyridine is far from a household name, but its influence runs deep in industries many of us rely on. Understanding its uses—and its risks—means supporting real-world solutions that balance innovation, safety, and environmental care. The way forward rests on putting health, transparency, and sustainability at the core of chemical progress.
3-Chloropyridine. Small name, big job in a lot of labs and chemical plants. Its chemical formula, C5H4ClN, boils everything down to its essentials—five carbons, four hydrogens, one chlorine, and a nitrogen stitched into a simple ring. Mention that backbone to any organic chemist, and you’ll probably get a tired but knowing nod. Pyridine rings, once you run into them, keep popping up in pharmaceuticals, weed killers, and even dyes.
Chemicals like 3-chloropyridine don’t typically become household names, even though they’re right at the heart of day-to-day life for the chemistry crowd. Walk through a synthesis project—say, trying to tweak a medication recipe—and there’s a need to swap around atoms in the right spots on a ring. That chlorine at position 3 shakes up the way the molecule reacts. Those little changes can deliver a totally new effect for researchers testing out a new crop treatment or a promising drug. In my own work brushing up on agrochemicals, I keep seeing 3-chloropyridine derivatives as building blocks. Turns out, adding a chlorine in just the right spot can transform a basic molecule into one that shrugs off insects or helps a new medicine slide into the right enzyme pocket.
It isn’t all chemical magic, though. With usefulness comes responsibility. Chlorine’s presence can mean extra hurdles for disposal and safety. Workers can’t treat 3-chloropyridine like some harmless kitchen salt. Spills or long-term exposure bring health questions. Pyridine on its own smells pretty awful, and chlorine compounds often remind us of lessons learned from industrial pollution. Strict handling rules protect both people and water. From factory to university lab, no one cuts corners on ventilation and protective gear.
Some folks think regulations stall innovation, but from where I sit, a better attitude changes that conversation. Open reporting, treating chemical waste as a problem to solve rather than a nuisance, and tinkering with reaction setups so less gets wasted—those help more than just ticking off a checklist. I’ve seen teams pool their knowledge to invent less hazardous by-products or come up with reusable catalysts, cutting the danger before it walks out the lab door.
Maybe what grabs me most is how something as simple as the placement of chlorine on a ring can merge cutting-edge research with old-school chemistry challenges. Ask the folks in pharmaceuticals, or those fixing up polluted sites, and you’ll hear the same thing: getting careless with these chemicals just causes headaches later. Consistent, honest education about risks, both among professionals and for newcomers, does more in the long run than any single rule set.
3-Chloropyridine might look like just another collection of atoms and bonds, but dig deeper and it’s a reminder. Every piece of progress in modern chemistry asks us to balance excitement with caution. Every choice—ingredient, process, or even what to teach the next generation—counts double when one molecule can mean so much.
3-Chloropyridine isn't a household name for most, but it comes up more than you'd think in fields like manufacturing, pharmaceuticals, and agrochemicals. I spent a few years surveying chemical storage rooms and lab benches—it's a solid contender for “most likely to be stored in a fume hood.” Anyone who's handled it understands why. Its presence offers plenty to talk about, not least because of the red flags it raises for health and safety.
Smelling this stuff even from a distance can set off alarm bells. It packs a pungent, irritating odor—hinting right away at possible respiratory trouble. Published toxicology studies paint a rough picture: the chemical can irritate eyes, skin, and lungs after short-term contact. Breathing it in or letting it touch your skin over time increases the risk of damaging effects, especially for lab workers or anyone involved in manufacturing processes.
My own experience echoes what chemicalsafety literature warns. Even small splashes demand immediate rinsing and a trip to the nearest eyewash. A colleague once underestimated how far droplets could travel, and learned the hard way with a nasty skin rash for a week. Backed up by resources like the National Center for Biotechnology Information, exposure management requires more than gloves; proper ventilation and face protection are basic essentials, not just nice-to-have.
One serious concern with 3-Chloropyridine is its classification as “toxic if swallowed or inhaled.” Acute exposure brings on nausea, headaches, dizziness, and breathing difficulties. Chronic exposure raises the stakes, possibly affecting the nervous system and major organs over time. Some animal studies point to liver and kidney stress at high doses.
There's another angle here rarely mentioned outside chemical circles—the ways this chemical behaves once it leaves the lab or factory. It’s moderately persistent in the environment, which means it sticks around long enough to pose a risk to groundwater and wildlife. This factor shifts the lens to a broader scope: human health and ecosystem health both feel the impact.
From my stint in research labs, nothing beats preparation. Preventing leaks and spills saves time, money, and hospital trips down the line. That means airtight storage, double-checking lids, and labeling every bottle. I’ve seen how quickly protocols slip when interruptions happen, only for an oversight to lead to a costly cleanup.
Workers and employers share responsibility here: regular training, up-to-date safety data sheets, and functional safety showers are just the starting points. Monitoring air quality in spaces where 3-Chloropyridine gets used or stored identifies problems before they turn serious.
Regulators in Europe, North America, and Asia have their say by listing 3-Chloropyridine under hazardous chemicals, and for good reason. Governments increasingly ask companies to substitute less harmful alternatives when possible. Green chemistry—developing safer, more sustainable chemicals—won’t change things overnight. But even now, scientists and engineers are working toward routes with reduced human and environmental tolls.
In the end, safety doesn't happen by accident. Careful handling, informed workers, and a real culture of safety make all the difference, whether in a big pharmaceutical plant or a small research lab.
The first time I worked in a chemistry lab, the thing that stuck with me wasn’t the glassware or the glowing reactions. It was the way everyone handled chemicals—especially ones like 3-chloropyridine. The label invited respect: flammable, toxic, volatile. Even outside a laboratory, these chemicals influence daily life, showing up in pharmaceuticals, pesticides, and even electronics. With their usefulness comes responsibility.
3-chloropyridine brings challenges because of its properties. It’s a liquid that catches fire easily, and inhaling its fumes turns a routine day into an emergency. The main lesson: always store it in a cool, dry place, far from anything that could spark. I have seen well-meaning but rushed colleagues put flammable bottles next to heat sources or forget to tighten a cap. Those moments turn into lessons none of us forget.
Facts matter. The flash point of 3-chloropyridine is low, so even a warm room or poor ventilation can invite trouble. Chemical compatibility runs through my mind when shelf space gets tight. You never want this stuff anywhere near oxidizers or acids; that’s a recipe for disaster. Real cases of fires in labs often start with two chemicals being too close in the dark corner of a storage cabinet.
Every chemical I’ve ever worked with slowly finds its way into the air if not sealed up tight. 3-chloropyridine evaporates at room temperature, so a loose lid can fill a work area with fumes. I remember a time the faint smell of pyridine lingered on my lab coat for hours—I hadn’t realized how quickly vapor spreads or how long it stays. That's why vapor-tight containers and proper ventilation systems aren't optional—they let us breathe easier and keep routine tasks safe.
A sharp label goes further than a fancy lock. Proper chemical names and hazard warnings signal caution. In every institution where I’ve worked, clear labeling cut through language barriers and prevented mix-ups. Segregating chemicals based on hazard class helped even more. Once, a cart loaded with incompatible solvents tipped over, but because bottles were segregated, nothing dangerous mixed or spilled.
No storage solution works without training. I’ve seen experienced researchers skip steps, thinking they’ve seen it all. Yet, basic habits—like inspecting containers for leaks and checking expiry dates—protected us. Routine checks reveal deteriorating seals or cluttered aisles. Fixing small problems early beats cleaning up a crisis.
A safer space starts with flame-resistant storage cabinets set away from direct sunlight, ignition sources, and acid cabinets. Proper spill kits sit on the shelf below because accidents happen. Well-ventilated rooms and regular airflow checks keep invisible dangers in check. It’s those simple steps that stop a normal day from turning into tomorrow’s headline.
Handling chemicals like 3-chloropyridine reveals how safety culture makes a difference. Good storage is built on facts, experience, and respect for the risk involved. It’s not about making things harder—it’s about making sure everyone goes home safe, every day.
Ask anyone who’s ever worked in a lab with tight budgets and stricter regulators — figuring out how long a bottle of 3-chloropyridine can sit on the shelf before it breaks down is more than a chemical trivia question. It impacts safety, research quality, and finances. In my years working around chemical storerooms, rumors and memory often dictated shelf life, not rigorous science, and that rarely worked out well.
3-Chloropyridine is a fairly stable compound, but nothing lasts forever. Exposure to moisture, light, or air speeds up degradation. The most obvious threat comes from hydrolysis — that’s the reaction when water sneaks in and starts snipping molecular bonds. If the seal isn’t tight, or the storage area sweats in summer, 3-chloropyridine can lose its punch. Sometimes degraded stock turns up through funky smells or color changes, but most complications quietly show themselves only after a crucial experiment fails or, worse, a hazardous byproduct forms.
Several trusted suppliers suggest a shelf life of about two to three years for unopened containers stored in cool, dry places, away from sunlight. That timeline never felt like gospel to me, because real-world conditions differ. Fluctuating temperatures in university storerooms or commercial settings chip away at a substance’s stability. Even simple errors like twisted caps and forgotten bottles in the wrong cabinet can shave months off usability.
No chemical lasts long when handled carelessly. I’ve seen well-sealed, climate-controlled bottles stretch well past a recommended expiration, and others spoil early due to a brief exposure on a bench. Most degradation accidents come down to how we store and handle the stuff. Investing even a little effort in proper storage pays off. For 3-chloropyridine, this means tightly sealed bottles, dry cabinets, and avoiding frequent opening. One-off mistakes, like using wet spatulas or forgetting to purge the airspace with inert gas, have cost more than a few groups months of work.
Chemical companies recommend periodic testing to confirm a reagent’s suitability, but tight budgets push that advice lower on the priority list. Thin-layer chromatography or spectroscopy checks might seem like a hassle, but they catch problems before they reach a flask. Over the years, I’ve lost count of experiments sabotaged by old or contaminated reagents. A quick check upfront would have saved hours.
Making a habit of labeling containers with receipt dates, opening dates, and periodic checks makes a world of difference. Training staff and students in chemical stewardship pays bigger dividends than just buying fresh chemicals on repeat. Lab management software or even simple spreadsheets have helped keep the shelf life question from turning into a guessing game.
Robust stock rotation and prompt disposal of expired 3-chloropyridine matter just as much as buying high-quality material to begin with. Shared labs often pool their leftovers, but if it’s unclear how long a bottle has been open, it’s a bigger risk than reward. Frugal as many labs are, gambling with degraded stocks costs more in lost productivity and safety risks than replacing questionable chemicals.
The discussion about the shelf life of 3-chloropyridine isn’t just about numbers or guidelines. It drives home the need for habits that value quality and safety. Investing in good training, storage, and routine checks goes a long way. In my experience, those habits do more to protect workers, budgets, and results than any amount of wishful thinking about expiration dates.