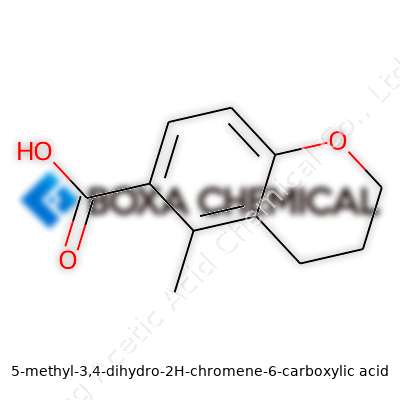
Chemistry thrives on curiosity and a steady, sometimes stubborn, dedication to trial and error. The discovery and study of 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid illustrate that point well. Over the decades, chromene derivatives pulled in attention for their complex aromatic systems and the promise of unique biological activity. Early researchers, likely working with glassware that seems archaic today, built a body of work exploring how slight changes to the chromene ring bring about new properties. The addition of a methyl group at the 5-position and a carboxylic acid at the 6 made this molecule stand out to folks chasing new pharmaceuticals and novel materials. From obscure syntheses buried in university journals to a molecule referenced in patents, the story of this compound runs alongside the broader growth of synthetic organic chemistry. Just as the field transformed with better separation methods and spectral analysis, interest in tailored molecules like this one grew, often shaped by needs in medicine, agriculture, and the search for sustainable materials.
The arcane names can put people off, but at the core, 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid stems from a simple logic—change a molecule ever so slightly, and sometimes the world responds in dramatic ways. On one end, it’s a research chemical with clear links to the chromene family, which includes compounds known for antioxidant and anti-inflammatory roles. In a practical sense, formulations drawing on this acid aim for a chemical scaffold that strengthens molecular stability or introduces fresh biological activity. Labs across the globe chase customizations like this, hoping to unlock something useful—a better treatment, tougher materials, or fresher crop protection. Commercially, it makes its appearances in specialty applications, but the real action happens in the lab, where properties get tested and new reactions start their journey.
Every synthetic chemist has memories of weighing out sticky yellow solids or watching crystals form in a flask. This acid, with its unique methyl twist and aromatic system, often crops up as an off-white powder or solid. Molecular weight, solubility profile, and melting point guide scientists in everyday handling—soluble enough in common lab solvents, stable in the air, but also reactive where it counts. The structure combines the stiffness of an aromatic ring with the potential flexibility from its dihydro-chromene portion. The methyl group and carboxylic acid aren't just decorations; they tune reactivity, making this molecule a handy tool both in synthesis and testing. It doesn’t sizzle with color under UV, but its response to reagents in the lab sparks excitement for those who know what to look for.
Serious research relies on precision, not just with glassware but with reporting. Scientists want to see a verified structure, clean spectral data, and a clear understanding of purity. Typical specs list molecular weight, chemical formula, and minimum purity levels—often above 98 percent. There’s sometimes a reference to standards such as IUPAC or CAS numbers to ensure everyone speaks the same language. Labs using this compound focus on safe handling and good labeling, not just because of regulatory expectations but because clear records let others reproduce work across borders and decades. Mislabel something and you waste weeks; get it right and research fits together.
Synthetic routes to 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid echo the growing sophistication of organic chemistry. Most start with chromene building blocks, tuning conditions with acids or bases, and choosing solvents that push reactions down the desired path. Common practice often relies on condensation or ring-closing steps, giving researchers control over selectivity and yield. In graduate school, I spent long hours hunting for minor changes—swapping out a catalyst or lowering the temperature—to boost conversion rates just one or two percent. Small optimizations multiply when you’re scaling up for further research. Purification, using chromatography or crystallization, leaves you with a product ready for the next steps, whether that’s testing directly or modifying for new experiments.
The fun doesn’t stop at synthesis. The methyl group might seem quiet, but chemists love tweaking positions to explore what changes. The carboxylic acid opens a gateway to hundreds of derivatives—from salts to esters and amides. Reactivity at the ring system invites oxidative modifications, hydrogenations, or even cross-coupling if conditions play along. In the hands of a creative chemist, this molecule becomes a launching pad. Previously, colleagues in the lab would exhaust the shelves hunting for reagents that unlock rare transformations, looking at this compound as a gateway to more complex structures. Every new derivative brings a chance for better biological data, improved stability, or a shift in the physical properties that put compounds into new application zones.
Chromene derivatives have a knack for picking up a list of alternative names, making literature searches a trial for students and seasoned scientists alike. Most chemists stick to the IUPAC name or recognize the compound through short tags like methylchromene acid. Across different suppliers or papers, expect to see variations referencing the position of the methyl group and the acid placement on the ring—a reminder that chemical communication sometimes juggles clarity with tradition.
Any seasoned researcher learns to respect every new reagent, and this holds for the chromene family. While not classified among the most hazardous substances, best practice means gloves, goggles, and proper ventilation. Standard data—LD50 readings, environmental impact notes, or storage advice—reach the desk of every lab technician. Fume hoods and good record-keeping limit accidents and let teams work with confidence. It’s not just about satisfying regulators; it comes from a culture built on learning that even the most familiar compound can carry a surprise. Laboratories driven by safety run smoother and attract collaborators who trust that work gets done responsibly.
Once this acid leaves the synthetic bench, its journey splits across a handful of promising territories. Its chromene core draws attention from pharmaceutical researchers hunting for new drug scaffolds, as changes here often lead to hits in early biological screens. These compounds navigate the line between innovation and risk as preclinical testing sorts the viable candidates from the rest. Agricultural scientists look for stable, reactive molecules for smarter pesticides or fungicides, and this chromene derivative fits the bill thanks to its mix of aromatic and acidic features. Material scientists, never to be left out, sometimes borrow from pharmaceutical logic, hoping to tweak thermal or optical properties in specialty polymers. For now, the mainstay remains early research, but the breadth of application points to potential expansion as new data emerge.
Every breakthrough ties to a wave of experimentation and dogged persistence. R&D on this molecule finds itself part of broader projects looking for next-generation drugs or smarter materials. Early cell tests might show promise—antimicrobial, anti-inflammatory, sometimes neuroprotective effects—which push groups to refine the structure and see what sticks in more complex models. Screening at scale calls for enough quantity, reliable purity, and a willingness to follow data wherever it leads, even if it means scrapping months of work for a better lead. Collaborations between chemists and biologists run thick in this field, as synthesis means little without the right biological partners. Teams learn, sometimes painfully, that one shift in position on a ring can mark the end or beginning of a research project. Funds trickle into the hottest application areas, and industry-academic partnerships help fill gaps, sometimes speeding up routes from bench to the world outside the lab.
Establishing the true toxicity of any fresh compound takes grit and patience. Even with promising early data, careful in vitro and in vivo studies tease out possible risks. This chromene acid, like many related molecules, draws scrutiny for its impact on cell viability, breakdown products, and environmental persistence. It’s not enough to clock a desired effect; long-term exposure and breakdown pathways matter if it will cross from the lab to real-world use. Older colleagues taught me to never trust a single assay, always double-check unexpected results, and to read between the lines when new data show up. A rigorous approach, across species and systems, limits risk down the line and helps regulators sleep at night. Any compound seeking approval must clear increasingly stringent safety hurdles, and rightly so.
It’s easy to overlook a single molecule against the churn of chemical progress, but 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid sits at the crossroads of several promising fields. Advances in drug design, green chemistry, and smart formulations promise more pathways to real impact. The steady improvement of computational methods and screening technologies gives researchers better shots at finding out where this compound fits best, from treating disease to helping build more responsive materials. Broader access to high-quality synthetic tools lowers the barrier for smaller labs and startups to chase new applications. Not every experiment will pay off, but compounds like this one remind us that a tiny tweak in structure sometimes opens doors that textbooks didn’t predict.
Ask any chemist about 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid and you’ll often see a look of recognition. This compound sounds complex, and it is, but its real-world use stretches far beyond the lab bench. It’s become increasingly important in both pharmaceutical development and synthetic chemistry, both fields that touch nearly everyone—whether you realize it or not.
Drug discovery always faces uphill battles: side effects, consistency, and how well a drug actually works. Here’s where unique ring structures like this chromene derivative come into play. Medicinal chemists use building blocks like this to create new drug candidates, testing tweaks in real time to see what sticks. The chromene core carries antioxidant and anti-inflammatory qualities, signaling promise for disorders ranging from heart disease to neurodegenerative illnesses.
Research suggests that the presence of the methyl group and carboxylic acid function offer easier sites for further chemical modifications. These tweaks can help drugs absorb better in the body or target specific organs more precisely. Scientists synthesize and test variants for use as anti-cancer agents or as new pain medications, building on this molecule’s biologically interesting backbone.
What often gets overlooked is how molecules like this help create even more complex chemical structures. Organic chemistry lives and dies by intermediate compounds that make reactions possible. The chromene family provides strong stability under a range of reaction conditions. Its methyl and carboxylic acid groups help direct new chemical bonds to just the right spot, which keeps reactions clean and product yields high.
If you’ve ever taken a chemistry course, you know the frustration of side products muddying results. Researchers often reach for this acid because it gives predictability—a rare blessing in bench science. This reliability boosts efficiency in flow synthesis setups that companies deploy for scaling up specialized drugs and specialty materials.
With promise, though, comes the need for oversight. Derivatives of chromene carry biological activity, but not always of the positive sort. Unchecked use in agricultural or environmental contexts could create persistent byproducts. History shows synthetic intermediates sometimes end up in waterways. Strict protocols and smart waste management practices belong in every facility using this molecule.
In my own experience in a university lab, even a bit of carelessness leads to safety incidents or environmental trouble. Chemistry departments hammer home the importance of responsible handlings, such as chemical fume hoods and careful disposal methods. Without upholding these habits, good science turns to bad outcomes—fast.
As the pace of medical innovation quickens, demand for flexible, reliable building blocks like 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid climbs. More institutions blend chemistry, pharmacology, and environmental science, exploring not just what these molecules can do, but how best to use them safely. Funding for greener synthesis, closed-loop waste systems, and better toxicity screens promises to keep risks in check while still banking on their full potential.
We owe much of tomorrow’s medicine and material science to thoughtfully chosen molecules developed right now. The chromene acid stands as more than a molecular curiosity. It’s a bridge connecting brilliant science with solutions the world truly needs.
Ask anyone buying just about any product—food, supplements, chemicals, even water—what matters most, and purity always climbs to the top of the list. That’s not by accident. Purity goes beyond a label or a number on a certificate. It shapes trust. No one wants to gamble on extra stuff lurking where it shouldn't be, especially when health or performance gets involved.
Growing up, I watched my grandfather turn away sacks of grain because someone slipped in odd-looking kernels. He’d lecture me about unmixed seed, how one scoop of something strange could ruin a recipe or yield. That lesson stuck. The purity of a product defines its value. Trust travels with it, and that trust isn’t won with marketing. It’s earned by proving you care about what’s inside—down to the last decimal.
Take pharmaceuticals. The United States Pharmacopeia lays out stringency so patients get what medicine promises, not a little more, or a little less, or a few stray milligrams of something not on the label. In food, the difference between 99.99% and 99.5% purity can mean the world for allergies and sensitivities—think peanut traces or gluten sneaking in where they’re not welcome.
Contaminants come from unexpected corners: unclean equipment, slipshod storage, or plain neglect. Even trace elements add up—lead, arsenic, microplastics—none belong in your meal or your pill. The World Health Organization keeps reminding folks: small lapses turn into big health issues, sometimes quietly, slowly.
Most people don’t stroll into a store, microscope in hand, ready to test flour, vitamins, or baby formula. Instead, we lean on manufacturers’ honesty and transparency. New technologies now let companies run even tighter checks: chromatography, mass spectrometry, ever-tighter benchmarks. Reputable brands publish test results or open their supply chains for review. Consumers aren’t asking for miracles, just accountability.
One trouble spot: not all regions have the same enforcement muscle. Some global markets carry products banned elsewhere. This isn’t fair, and it’s dangerous. Too many stories fill the news—tainted medicines, mislabeled foods—that end with suffering, hospital stays, or long-term health harm. Even with watchdogs in place, fakes and shortcuts snake through supply lines every year.
Buyers want a clear answer to a simple question: “How pure is this?” That means sellers have a real duty to test, to fix problems before products ever see a shelf, and to share findings openly. Real transparency wins repeat customers, and it keeps brands out of the headlines for the wrong reasons.
Individuals can get smarter, too. Scan labels, look up batch numbers, read third-party reports instead of relying on the front of the package. Support outfits that support rigorous testing and don’t shy away from explanation. Even small changes in purchasing habits make ripple effects across industries.
No quick fix will sweep away all contamination troubles. Still, emerging ideas show hope. Blockchain in supply chains, publicly posted audits, and stricter penalties for deception start shifting power back to everyday buyers. The idea: purity shouldn’t be a privilege for the few who can afford boutique brands. It ought to be the bare minimum.
If everyone, from farmers to scientists to folks looking for groceries, keeps pushing for truth and transparency, purity won’t stay just a technical term—it’ll become a promise delivered.
Anyone who’s spent time in a research lab knows how easy it can be to take shortcuts with chemical storage. Unfortunately, even a moment’s lapse can mean ruined experiments, a hazard to your health, or even a costly cleanup. The world of organic chemistry is littered with stories about compounds gone bad on a forgotten shelf. Thinking specifically about 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid, experience teaches that proper storage isn’t just a best practice—it’s the only way to work safely and keep your work reliable.
This compound stands as a solid organic acid, and like many organic acids, it cringes at uncontrolled heat, ambient humidity, and the wrong kind of light. Through my years in both academic and commercial labs, I’ve seen more than one bottle of precious intermediate degrade into uselessness because it ended up near a window or on a warm shelf. Humidity isn’t any kinder. Even a small desiccant packet inside a bottle cap can save your sample from drawing in enough moisture to throw off a precise reaction down the line.
For chemicals like this, you want a cool, dry spot—a place where temperature swings stay limited and sunlight never intrudes. A standard flammable materials cabinet often gets used for anything organic, but with a solid carboxylic acid, what really counts is the basics: avoiding light, skipping high-moisture environments, and keeping temperature stable.
I once stored similar acids in glass bottles with tightly sealed caps, packing a silica gel sachet inside for good measure. On cramped shelves, clear labels and a logbook cut down on both accidents and wasted effort. Reliable storage supports reproducibility. There’s something reassuring about opening a bottle months after you last touched it and finding your material in exactly the state you left it.
Loose protocols and vague habits almost always end badly in a lab. Proper chemical storage isn’t about hoarding bubble wrap or obsessing over locked cabinets. It’s about upholding a culture of trust in the workspace, where everyone looks after the integrity of their own reagents and, by extension, watches out for one another’s well-being. Every chemist who has reached for a tub, only to find themselves holding an unreadable label or a rehydrated mess, knows why these details deserve attention.
Mislabeled, degraded, or contaminated compounds stall projects and can set off a domino effect of bad data and questionable conclusions. Consistent, clear storage helps make repeatable, credible results possible—something no flashy safety poster can substitute for.
The fix isn’t complicated. Store 5-methyl-3,4-dihydro-2H-chromene-6-carboxylic acid in a dry, cool, and dark place. Use airtight containers. Mark the date you opened the sample, and keep tabs on any signs of clumping, discoloration, or odd smells. If equipment for environmental controls is available, use it, but good labeling, dedicated spaces, and informed colleagues make the biggest difference. The strongest labs aren’t just the ones with the best gear—they’re the ones with habits that protect both people and results.
It’s easy to forget how many different chemicals move through labs, factories, classrooms, and warehouses each day. I’ve seen experienced chemists reach for a vial assuming that something familiar-cased as benign, only to realize later that no one checked the safety sheet. That’s not an uncommon scene. Without pausing to check for safety data, trouble finds an open door. The reality is, the search for an MSDS—those Material Safety Data Sheets—too often happens after a spill, a splash, or someone coughing in the hallway.
It’s tempting to think, with computers and smartphones everywhere, up-to-date chemical information would always be on hand. Yet, missing paperwork or outdated files still pop up. The Occupational Safety and Health Administration (OSHA) in the US and similar bodies in other countries steer employers to keep material safety data sheets within easy reach. These aren’t just pieces of paper; they break down real hazards—fire, poisoning, reactivity—that affect daily routines. I remember a colleague burned after cleaning a benchtop with an unfamiliar compound. No information packet, nothing but guesswork. That scene ended with an emergency room visit and an ugly surprise about compatibility with water.
An MSDS tells you more than just toxic effects. It spells out storage needs, required gloves, what to do if someone swallows a compound, and what type of container won’t explode. The difference between a bad scare and a minor mess comes down to knowing details. Organic solvents, industrial salts, custom-blended powders—each brings its own quirks. Over the years, I’ve watched people skimp on training, trusting their gut over the fine print, but that trust rarely pays off. Benzene’s vapor creeps into lungs before you notice the smell. Hydrofluoric acid—odorless and sneaky—can damage tissue deeper than you might think.
Many new compounds hit the market or float around research labs each year. Safety documents lag behind. Specialty chemicals from small suppliers sometimes ship without thorough documentation. In my own consulting, researchers use hand-mixed reagents made on the fly with little background. Official agency requirements exist, but loopholes remain, and the pressure to innovate often outweighs old-fashioned record keeping.
The fix starts with an expectation that every chemical, no matter how obscure, travels with a clear safety sheet. No one should rely on an internet search after an incident has already happened. Regulatory agencies must push hard for suppliers—large or small—to prioritize updated, digital MSDS access and plain-language guides. In labs and companies, regular training works better than checklists and audits alone. I often point out that sharing a printed-out MSDS at the start of the week can save not only skin but entire projects from derailment. Letting everyone see the worst-case scenario sharpens caution more than any sign on the wall.
Chemistry built the modern world with astonishing speed. That same speed lands people in the hospital if corners get cut. There’s no secret to avoiding tragedy, just a willingness to double-check, question what’s missing, and demand clear answers from suppliers. If the safety data isn’t available, the work can wait. No experiment, batch, or process runs smoothly for long without it.
Anyone curious about what’s truly inside a supplement, a bag of flour, or even a skincare cream will likely find peace of mind through a certificate of analysis. In short, this document shows exactly what’s in the product, backed by third-party or in-house laboratory testing. I remember getting curious about some herbal capsules I was buying online. Claims looked great, but I found myself asking: how can I know I’m actually getting what I pay for?
Digging deeper, I learned that a certificate of analysis, or CoA, exists for this exact reason. It’s not just a technical report. It's a window into ingredient purity, potential contamination, and whether a product holds up to its promises. These certificates matter most to people like me who care about quality, especially those with allergies or specific dietary restrictions.
Stories in the news have shown supplements spiked with undeclared substances, food powders tainted with heavy metals, and cosmetics hiding more than they declare. A CoA can flag these problems before they reach the customer. People shouldn’t have to gamble with their health or trust slick marketing. The recent surge in wellness brands, especially the flood of unregulated internet shops, shows there’s more need than ever for transparency.
Skilled workers in labs test batches for pesticides, heavy metals, bacteria, and ingredient levels. I spoke once with a quality manager at a small supplement company who described the stress and cost involved, but the relief in knowing each lot matched the label. Certificates often catch out-of-spec batches, and lots that don’t match up do not end up in stores—or at least, that’s how it’s supposed to work.
Whenever I buy coffee, protein powder, or even CBD oil from an unfamiliar source, I look for evidence that the producer knows exactly what they're selling. A business that shares CoAs straight away has nothing to hide. The opposite raises suspicion. Transparency here isn’t just about compliance; it shows who respects the customer and who sees them as a transaction.
Regulatory rules differ between regions. In the United States, the FDA doesn’t pre-approve dietary supplements, so the onus falls on companies to ensure products are clean and accurately labeled. Europe and Australia have stricter rules for certain items, but loopholes remain, especially for products bought online. This fragmented approach puts the burden on the customer to ask tough questions.
Some companies put QR codes on packaging so shoppers can pull up test results on the spot. Others share full CoA PDFs on their websites, sometimes even batch-specific documents. Still, transparency remains voluntary in most markets. Companies unwilling to share these documents often have something to hide, or are simply cutting corners.
I feel more at ease handing my money to a company with a proven commitment to safe and honest products. Giving customers real access to certificates of analysis shouldn’t feel revolutionary; it should be expected. If enough people keep asking, it can push more brands to make honest information the default, not the exception.