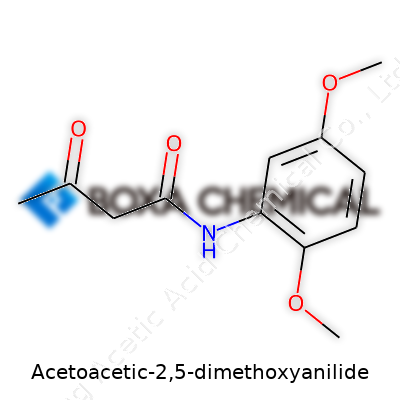
Acetoacetic-2,5-dimethoxyanilide didn’t arrive overnight. Exploration of acetoacetic derivatives reached a serious stride in organic chemistry labs throughout the late 19th and early 20th centuries. Chemists kept building on carbanilide frameworks, looking for tweaks that might bring out new colors, functions, or medicinal promise. The itch to modify anilide moieties led researchers to the methoxy-substituted versions, and work with acetoacetylation grew. Universities, industrial research arms, and lone scientific minds dug into synthesis, handling noxious-smelling amines in glassware that demanded care and skill. As catalogues of chemical structures expanded, Acetoacetic-2,5-dimethoxyanilide found its way into journals, patents, and industry manuals. I’ve seen how these old syntheses, powered by trial and error, paved the way for today’s more refined, sometimes automated, chemical processes. This kind of progress didn’t just live on paper—it inspired whole generations of chemists to tinker and question what was possible with aromatic amides.
Mention of Acetoacetic-2,5-dimethoxyanilide often lands in specialty dyes, pigment intermediates, and analytical chemistry circles. The core draws from its molecular structure: plenty of reactivity from the acetoacetyl side and stable electron-donating power from the two methoxy groups. I’ve watched researchers use it for synthesizing specialized azo dyes, laying groundwork for lasting colors in textiles and plastics. Demand hasn’t just come from curiosity about how it works, but from what it does—delivering reliability in applications where fine-tuned performance matters. The story isn’t about filling a shelf in a laboratory—this compound’s journey from basic research to practical functionality tells a lot about how chemical innovations underpin everyday tools.
Hands-on chemical work offers a straight-up view for evaluating a substance. Acetoacetic-2,5-dimethoxyanilide generally appears as a pale powder; its solubility and melting point can give researchers clues to purity and reactivity. That slight, sometimes nutty odor reminds one of many aromatic amide compounds. Chemistry students may grouse about keeping it dry, but the tendency of some acetoacetyl derivatives to absorb moisture or slowly break down underscores the need for suitable storage. Chemical reactivity hits home most when handling derivatives. Mixing it with acids or oxidizing agents isn’t trivial. Broadly, the methoxy groups alter electron density in the aromatic ring, tuning the reactivity profile for downstream processes—something I first appreciated after comparing reactions with unsubstituted anilides under identical conditions and watching the color changes hint at underlying orbital differences.
Chemical supply chains owe safety and utility to solid documentation. Specification sheets for Acetoacetic-2,5-dimethoxyanilide routinely flag minimum purity, allowable trace contaminants, and recommended containers. The extra value, though, comes from clear labeling that walks users through main hazards and special notes on disposal or accidental contact. Having worked in labs that saw confusion over lookalike bottles, I can’t stress enough how wrong things go without rigorous labeling. Mistaking an amide for an acid in a synthetic protocol wastes time and could spark more serious issues. Regulations and standards—both local and international—filter down into the technical forms, leading to batch-level barcodes and safety warnings that meet the current best practice instead of leaving things to chance or improvisation.
Making Acetoacetic-2,5-dimethoxyanilide involves organic reactions that go back generations, yet every chemist who’s tried it learns something new about reaction control. The favorite method starts by condensing 2,5-dimethoxyaniline with an acetoacetylating agent. Overheating, incomplete mixing, or contaminants change outcomes: cleaner, smoother reactions need patient monitoring and pure starting materials. I remember a time a colleague, eager to squeeze out more product, pushed the temperature too far, producing tarry byproducts that cost us the whole day. Small variations in solvent, temperature, and reaction time aren’t just detail—they shape yield and purity dramatically. Post-reaction work, including washing, filtering, and sometimes recrystallizing, rounds out the job, and doing it right brings real satisfaction to anyone who values hands-on synthesis.
Once you’ve got it, Acetoacetic-2,5-dimethoxyanilide offers rich avenues for further chemistry. That acetoacetyl group can undergo condensation with hydrazines or amines, setting up for various heterocyclic structures popular in dye chemistry. Electrophilic aromatic substitution gets a twist from the methoxy groups, which open up unusual substitution patterns compared to simple anilides. Oxidation, reduction, or coupling reactions—each draws out a different aspect of the molecule’s character. Experiencing the thrill of a successful coupling, followed by an intense pop of color during azo dye formation, stands as a reminder of just how important these reactive points can be for building complexity step by step.
Walk through chemical catalogues and the names pile up: 2,5-dimethoxyacetoacetanilide, 2,5-dimethoxyphenyl acetoacetanilide, even systematic IUPAC versions that stretch for sentences. Each name tracks a slightly different local convention or commercial preference. Old-style names stick in some labs, modern “clean” nomenclature dominates in regulated settings, and anyone ordering or cross-referencing batches learns quickly how misunderstandings creep in. I’ve met researchers frustrated by near-identical names, only to realize a methyl versus methoxy slip wasted grant money on the wrong chemical. The bottom line: clear and consistent communication matters as much as any synthetic yield.
Day-to-day chemical work builds routines based on practical experience and published guidelines. Acetoacetic-2,5-dimethoxyanilide joins a long list of substances where appropriate gloves, goggles, and fume hoods protect those at the bench and those cleaning up. Mistakes—spills, poor ventilation, ignored expiration dates—never feel minor. I remember one post-doc who learned too late that certain acetoacetyl derivatives could irritate skin or mucous membranes, especially in warm, humid weather. Waste disposal follows strict rules, reflecting growing concern about both human safety and environmental burdens. Some labs also focus on limiting solvent waste from purification, echoing an industry-wide trend towards green chemistry. The most forward-looking safety cultures treat standards not as boxes to check, but as habits that make the work sustainable for researchers and everyone downstream.
Most interest in Acetoacetic-2,5-dimethoxyanilide comes from specialty dye synthesis, but its intermediate status leads to overlap with pharmaceuticals and materials chemistry. The compound plays a key role in building vivid, lightfast colorants used in fabrics, plastics, inks, and lake pigments. Industries looking for tailored hues and functional coatings lean into what this compound and its derivatives bring: chemical handles to adjust final properties by molecular design. Researchers see its structure—a blend of reactive and protecting groups—as perfect for crafting new heterocycles sometimes useful as pharmacophores or diagnostic agents. From my own perspective, seeing its fingerprints across analytical chemistry techniques and in advanced organics courses kept me aware of the breadth of its impact.
The story of Acetoacetic-2,5-dimethoxyanilide in labs is far from finished. Innovation comes from scientists eager to expand the range of colors, stabilities, or reactivities that this molecule enables. I’ve talked to collaborators using it as a scaffold for fine-tuning the absorption or fastness of new dyes, while another group targets modifications that could cut synthesis time or toxic byproducts. Green chemistry pushes have already led research teams to swap out noxious solvents, reduce energy input, and recover more byproduct streams. A big piece of the puzzle involves polymer chemists trying to link these structures in new coatings or functional thin films. Advances in analytical measurement—NMR, GC-MS, crystallography—mean fresher data and broader insight, letting science move faster without sacrificing the reliability that comes from old-fashioned methodical checking.
No one wants surprises from chemical exposure—especially after headlines about poorly understood risks of industrial compounds. Toxicity studies with Acetoacetic-2,5-dimethoxyanilide look at both acute and chronic effects in lab animals or cell cultures, assessing skin and eye irritation, cytotoxicity, and environmental persistence. Anecdotes circulate about researchers who learned the hard way to avoid direct contact and respect disposal rules. What stands out across regulatory guidance is the call for careful personal protection, strict waste stream monitoring, and better data on downstream metabolites. As scientists uncover metabolic fate and breakdown pathways, safer handling protocols gain nuance and precision, aiming for a balance between productive use and responsible stewardship across supply chains.
The push to innovate in color chemistry, specialty polymers, and advanced diagnostics drives steady research into derivatives like Acetoacetic-2,5-dimethoxyanilide. Sustainability concerns mean looking at how to make, modify, and dispose of chemicals with less waste and lower hazard. Ongoing projects evaluate biobased feedstocks for starting materials, improved purification without halogenated solvents, and scalable routes that sidestep hazardous intermediates. Growing use in screening for new pharmaceuticals inspires parallel safety and biodegradation research. Industry and academic collaboration shapes the next generation of bench-to-business work, guided by lessons from past syntheses and forward-looking risk management. Communities who rely on dyes and materials made from chemicals like this have a stake in supporting research that tunes quality and safety without closing doors to creativity or innovation.
Whenever I see a burst of orange or yellow from synthetic dyes, I think about the unsung helpers behind the scenes. Acetoacetic-2,5-dimethoxyanilide, with its tongue-twister name, works quietly in the background, powering the dye that ends up in the world’s garments, plastics, and printing inks. Many folks don’t realize that everyday products often owe their vibrancy to fine-tuned chemicals like this one.
Growing up, my family owned a screen-printing shop, churning out t-shirts in every color imaginable. Running the press, clogged screens turned a simple job into a nightmare, which often boiled down to the stability and solubility of the dye. Acetoacetic-2,5-dimethoxyanilide steps in as a coupling component—an essential part of the chemistry that creates azo dyes, especially those rich yellows and oranges that remain vivid after many washes.
In chemical terms, it acts as a building block for these dyes, letting manufacturers produce colors that hang on through heat, sweat, and sunlight. It’s easy to take this for granted, but before the right compounds came along, fading and off-color prints were the norm. Strong, reliable color brought consistency to manufacturing, which pushed synthetic dye production far past anything natural pigments could offer.
The story doesn’t stop at t-shirts. Acetoacetic-2,5-dimethoxyanilide finds its way into plastics, food packaging, and even leather products. Its ability to create “fast” color, resistant to both light and washing, comes from tightly controlled reactions in the dye factories. That means plastic buckets or vibrant food wrappers stay the same color for months, sometimes years. Stability matters not just for looks, but for safety—nobody wants a food label leaching chemicals into dinner or faded safety instructions on packaging.
Looking at trade data, synthetic dye production comes with pitfalls. Dye pollution is a dirty secret, as some production methods release toxins into waterways. Communities downstream from dye plants know this story too well—contaminated water, sick fish, and high cleanup costs haunt those dealing with old-school processes.
Change has started happening. Companies have invested in closed-loop manufacturing, which captures and reuses the chemicals that usually slip through to rivers and lakes. Rigorous testing now screens new dye components for safety before they leave the lab. Global chemical watchdogs, like the European Chemicals Agency, use updated toxicology data to help keep workers, consumers, and the environment safer.
The future of dye chemistry, including workhorses like Acetoacetic-2,5-dimethoxyanilide, rests on balance. Industries crave color that lasts and processes that don’t wreck the neighborhood. The push for transparency, real accountability, and smarter chemistry is growing stronger every year, driven by both regulation and consumer demand.
Every time attention shifts to what’s in the dyes behind common goods, a window opens for meaningful change. The world’s not about to give up color—most folks like their t-shirts bright and their packaging bold—but with the right choices, it’s possible to keep those hues vivid without leaving an environmental bill for the next generation.
Not every chemical you run into in the lab asks for the same kind of respect, but Acetoacetic-2,5-dimethoxyanilide sits high on my personal list. I’ve learned over time that certain compounds will not forgive a Sunday-afternoon attitude. This one, a mouthful only chemists seem to love, has qualities that call for attention: it isn’t a common household name, but it sure carries real risks for skin, eyes, and—perhaps worst of all—lungs.
Information from longstanding chemical databases such as PubChem and strict lab safety manuals point out that aromatic anilide derivatives have toxicity that can fly under the radar. Inhaling the dust or even skin-level contact sometimes slips by unnoticed, but effects can follow hours later. Anilides often provoke allergic reactions or something more persistent. Not something you want on your conscience or your hands.
Let’s get practical. I’ve seen too many students grab powders barehanded. For acetoacetic-2,5-dimethoxyanilide, gloves matter—a lot. Nitrile gloves stand up better than latex, especially since some solvents used with this chemical bully latex into tearing. Lab coats, fastened cuffs, and sleeves always rolled down keep your arms from brushing up against wayward dust. Safety glasses don’t leave your face, even if the reaction mix seems “quiet.”
Well-ventilated space isn’t just a good habit. Strong exhaust hoods pull airborne bits and vapors where they belong—away from lungs. If a lab runs without good airflow, I step out. Chemical exposure through air feels sneaky but does most of its work without you knowing. You’ll want dedicated spill pads and neutralizing agents close—preparedness beats panic.
Sifting through a safety data sheet means more than ticking boxes. Manufacturers publish bare minimums to stay within legal lines, but real safety grows from reading between those lines. The half-life of breakdown products, data on chronic exposure risks, and whether heat or light speed up reactions deserves your attention. Chemicals like acetoacetic-2,5-dimethoxyanilide sometimes break down into more dangerous cousins after a few months in a hot storeroom.
Don’t let “not explosive” or “not highly flammable” lull you. I’ve watched stable powders morph into a mess after a bottle seal fails or as labels fade. Allergy potential increases sharply for some lab workers, particularly after repeated exposure.
Building a culture that values safety is more than rules on a wall. I’ve found the best labs run on clear communication. Everyone announces chemical transfers out loud, not just with sticky notes stuck to the hood. Full label details, including prep date and initials, help hold people accountable. If anyone feels a spill’s been covered up or something’s missing from the safety gear, open discussion follows—without pushback from supervisors.
Routine training partnered with open-door reporting keeps risks in check. Eyes-only training videos never replace hands-on practice. Review of chemical storage—away from acids, oxidizers, or sunlight—reminds everyone that carelessness often hides in the mundane.
With acetoacetic-2,5-dimethoxyanilide and its kin, safety doesn’t happen on autopilot. It starts with people willing to slow down, ask, and think twice, even when hurrying feels easy. I wouldn’t call it fussiness—just the way to walk out healthy after a long day in any real research lab.
Standing in a college laboratory years ago, I learned that chemistry shows up in all sorts of places, not least in the names that chemists dream up for their discoveries. Acetoacetic-2,5-dimethoxyanilide isn’t a household term, but its structure brings together some familiar building blocks seen across dyes, pharmaceuticals, and chemical research. If you can break down its name, the chemistry opens itself up.
The structure blends an acetoacetic group – think of it as a four-carbon backbone with a double bonded oxygen (a ketone) and an extra oxygen that’s part of an ester or amide linkage. Hitching that to a benzene ring, which carries two methoxy groups at positions 2 and 5, brings both stability and a way to shape its chemical behavior. Toss in an anilide linkage — a type of amide where nitrogen sits connected to a benzene ring — and you end up with a molecule whose pattern repeats in several industrial colorants and specialty chemicals.
In industry, the little tweaks in chemical structure make or break a compound’s value. The methoxy groups change how electrons swirl over the ring, giving the whole molecule new interactions with solvents, acids, and light. In real-world applications, chemists pick molecules like this for their resilience and how well they anchor in polymer chains or interact with biological systems.
I’ve seen similar chemicals step in as intermediates in organic synthesis. Their predictable reactivity helps keep reactions under control and waste at a minimum. In dye chemistry, rings dressed up with methoxy groups often produce stable colorants that hold their color longer and put up a fight against fading and washing out.
With any synthetic chemical, especially one that can be found in dyes and specialty polymers, questions always come up about impact beyond the lab. Methoxy-substituted aromatic amides can linger in the environment. Regulations now push for transparency in sourcing and handling – not just because it looks good on paper, but because water treatment plants and soil bacteria sometimes struggle to break them down. From ECHA databases, we know that molecules with robust aromatic backbones survive many natural breakdown processes.
There’s also human health to keep in mind. Even a harmless-looking compound can turn risky in the wrong setting. Inhalation or direct skin contact with powder dyes—containing similar structures—has landed workers with respiratory and sensitization problems over decades. That experience drove responsible manufacturers toward implementing enclosed handling systems and better personal protection protocols.
Piecing together safer alternatives and better processes sits atop many chemist’s agendas. For acetoacetic-derived chemicals, moving toward more degradable side chains, less persistent ring structures, or using bio-based feedstocks means less environmental strain. Some researchers are retooling classic synthetic routes, using enzyme catalysts or solvent-free conditions to keep things cleaner, drawing on years of practical lab experience to drive down waste and improve yields. As societies wake up to chemical persistence and exposure, these shifts matter more than ever.
Understanding chemical structure isn’t just a textbook exercise — it shapes what ends up in our water, our workspaces, and our day-to-day lives. Better knowledge leads to smarter design, safer products, and new opportunities for innovation.
Anyone spending time around organic compounds knows that safe storage isn’t just a science class lesson. Acetoacetic-2,5-dimethoxyanilide stands out because it’s not a bottle you just stick in a cupboard and forget. Over the years, labs and workplaces have learned that a minor slip-up—leaving a cap loose, ignoring a humidity spike, crowding shelves—can turn a stable chemical into a disaster. Stories spread quickly among lab techs: a little ignorance ends with everyone blaming the “unexpected.” Truth is, poor handling mixes with bad storage much more often than you’d think.
Acetoacetic-2,5-dimethoxyanilide, like most anilide derivatives, has this knack for changing its character when environmental control goes out the window. Heat kicks off breakdowns; moisture invites lumps, or worse. In my own work, opening a container after months in a humid storeroom always led to clumped or yellowed powder—pretty good signs you’re dealing with more than a simple inconvenience. Chemical suppliers have documented that even mild temperature swings speed up decomposition, which bumps up risk, waste, and costs.
People sometimes shrug at dry, cool, well-ventilated spaces, but there’s a reason every chemical handbook keeps repeating those three conditions. Temperature control helps the compound stay true to its original state. Low humidity stops water-loving reactions from getting started. Good air flow means fumes don’t gather, while, more importantly, oxygen doesn’t linger inside containers, kicking off unwanted reactions. It’s hard not to sound preachy, but this basic care keeps everyone safer and stops product from going bad long before its shelf life is up.
The choice of storage jar or can makes a bigger difference than some realize. Some shops cut corners by transferring chemicals into generic bottles, not seeing the link between a loose-fitting cap and a strange smell seeping into the aisles. For acetoacetic-2,5-dimethoxyanilide, glass with tightly sealed tops tends to top the list. It handles most interactions, blocks stray oxygen, and survives the occasional bump better than plastic. I’ve also learned to label clearly—not just for me, but for the next person, who might not know what to look for when picking up a container.
Ignoring good storage habits causes a slippery slope of trouble. Instead of white or off-white powder, a degrading chemical discolors, sends out odd odors, or loses effectiveness—setting off alarms for both researchers and safety officers. Industrial processors sometimes find the costlier lesson: bad batches bring back product complaints, equipment issues, or the headache of regulatory visits. Records from chemical safety agencies point to storage mistakes as a top reason behind many contamination scares, proving this isn’t just nitpicking.
Better storage depends less on high-tech systems and more on good habits. Checking that seals fit, tracking temperatures with affordable loggers, rotating stock before it turns old—each step saves headaches. For bigger shops, clear training manuals help new workers avoid rookie mistakes. Some companies use humidity packs or dedicated climate cabinets, a bigger investment than a cardboard box, but still far cheaper than dealing with wasted chemicals. At the end of the day, steady routines prove worth their weight in preserved compounds and fewer safety incidents.
Products like acetoacetic-2,5-dimethoxyanilide rarely turn up at the kitchen table, but step into a coatings or specialty chemical lab, and the story changes. Whenever I visit one of these places, I pay close attention to two things before anything else—what the stuff looks like, and how pure it is. That’s not about formality. In my own lab work, these qualities can decide whether a batch turns out as a winner or flops before it leaves the flask.
Talk about purity, and most people think about high grades and precise percentages. For acetoacetic-2,5-dimethoxyanilide, purity often means making sure you’re not buying a product that brings in trouble. Impurities love to ruin reactions or stain finished products. When a batch of pigment or dye comes in even a notch below spec, customers notice defects—sometimes in color strength, sometimes in stability. Over the years, even tiny differences have cost more than one chemist a weekend fixing what should’ve been right from the start.
Lab insiders want purity numbers above 98% for starting compounds like this. Less and you’ve invited a chemistry wild card. Anything that claims “high purity” but turns up cloudy or with odd flecks—send it back. Solid science stands on the details that eyes and instruments both can spot. There’s no flex on that.
Chemicals talk, even when they’re silent. With acetoacetic-2,5-dimethoxyanilide, the expected appearance matters as much as what shows up on a COA. Chemists expect a powder, off-white to very pale yellow. Going much darker means something happened along the way—left out too long, exposed to a little moisture, or handled with dirty gloves. That might sound picky, but nobody wants a product that started to break down before it even had a chance to perform.
I’ve seen years of inconsistent samples, and the consistent lesson is this: anything slumping toward tan or brown isn’t going to make customers happy. Chips, caking, or clumps tell another story—one about poor storage or a rushed drying step. These clues show up before anyone grabs a spectrometer, and they help avoid surprises down the road.
Experience taught me that only tight process controls and mindful suppliers keep the bar high for appearance and purity. Relying on a trusted supply chain keeps products free of the nagging impurities that mess up downstream work. Reputable labs don’t skimp on the basics—HPLC, melting point checks, and even old-fashioned visual tests. These days, you find cross-checks like IR scans or TLC plates, too. Their goal isn’t box-ticking; it’s making sure nothing slips past.
Keeping that focus, the industry keeps an eye on global safety and quality standards. Organizations like ISO and ASTM push for stricter purity checks, which can only help customers and consumers alike. Every shipment that matches expectations—visually and on paper—saves time, money, and sometimes credibility.
A long career in labs showed me that talking about purity and appearance isn’t about politics or red tape. It’s about trust. Whether you make coatings, build a medicine, or craft a new color, every material tells a story—and you want one with a happy ending. Attention to appearance and purity doesn’t just satisfy inspectors. It leads to results that work, batch after batch.