The field of organic chemistry has never stood still, and every new compound brings both curiosity and challenge. N,N'-(1,4-Phenylene)bis(acetoacetamide) didn’t spring up from nothing; it reflects decades of research in aromatic chemistry and the pursuit of novel bis-amide compounds. Researchers in the late 20th century started examining acetoacetamide derivatives more closely for their unique reactivity and structure. A molecule marrying the straightforward benzene ring with two acetoacetamide groups seemed almost tailor-made for versatility. Through the years, greater understanding of reaction mechanisms, advances in purification, and improved detection methods have all contributed to making this molecule accessible for a broader audience. The academic push for multifunctional intermediates found a good candidate here, and the compound’s profile continues to grow in both laboratory and applied settings.
At first glance, the molecule’s name looks complex, but the structure tells a simple story: a benzene ring pops at the core, with two acetoacetamide groups anchored at the para positions. It sits at a sweet spot for building larger, more intricate molecules. Having both nucleophilic and electrophilic sites, it often steps into multi-component reactions or serves as a platform for generating heterocycles and other drug-like scaffolds. The dual acetoacetamide units also make it handy for cross-linking or chelation, areas chemists and material scientists keep returning to.
This compound rolls out as a solid at room temperature. Its color tends to lean off-white, sometimes yellowish, depending on impurities. Thanks to the amide groups and the aromatic nature, solubility patterns matter: soluble in polar organics, less so in basic water or nonpolar solvents. Its melting point comes in reliably at the high end for organic solids, reflecting the stable intermolecular forces in play—hydrogen bonding through the amide hydrogens and electron-rich rings. Chemically, its acetoacetamide side arms open doors for enolization, condensation, and addition, making it an attractive cornerstone for researchers cooking up more complex products.
Reading technical specs might seem like a numbers parade, but quality matters more than digits to most lab workers. Purity levels shape everything downstream—a trace impurity could throw off a catalyst or skew results in pharmaceutical screens. Labels try to strike a balance: providing enough information to ensure safe handling, storage at cold, dry, and dark conditions, and guidance for using the compound in scale-up or more delicate synthetic routines. Researchers appreciate clear marking of batch numbers and expiry, since the compound can undergo slow hydrolysis if left exposed for long enough.
Making N,N'-(1,4-Phenylene)bis(acetoacetamide) teaches a few lessons. Starting materials usually feature 1,4-phenylenediamine and ethyl acetoacetate or a similar acetoacetate ester. Reaction steps follow classic organic tricks: usually condensing in ethanol or methanol, assisted by mild base catalysts to push the nucleophilic substitution. The workup involves repeated washing, crystallization, and filtration, demanding patience and skill. Scale-up brings headaches as yield, purity, and cost have to balance. Plenty of chemists keep notes on process tweaks to tackle batch variability, illustrating how each lab’s experience shapes their relationship with the compound.
Versatility stands out as a defining trait here. That central phenylene bridge holds its acetoacetamide wings ready for a range of reactions. The active methylene in the acetoacetamide units jumps into condensation chemistry—think Knoevenagel or Hantzsch-type reactions—without missing a beat. The amide nitrogens, though less reactive, can become targets for electrophilic modification. Chemists often use the compound as a synthon for new heterocycles, bridging to fields from medicinal to coordination chemistry. Coupling reactions, complexation, and even metal-organic frameworks increasingly look to this molecule as a unique building block.
Names carry weight in chemistry. This molecule pops up under several guises: 1,4-phenylenebis(acetoacetamide), para-bis(acetoacetamide)benzene, and other analogs depending on language and naming conventions. In some catalogs, a shortened derivative code might show up. The collection of names sometimes causes headaches for researchers scanning databases, so awareness of these synonyms saves time and prevents unnecessary reordering or duplication.
Working with N,N'-(1,4-Phenylene)bis(acetoacetamide) doesn’t typically raise alarms, but reasonable caution stays the course. Dust from fine powders irritates eyes and skin, and some ammonium byproducts can continue forming after initial synthesis. Good ventilation, gloves, and the standard set of personal protective equipment handle most situations. Proper storage extends shelf life and reduces the risks of slow decomposition or unwanted crystallization changes. Disposal falls under organic chemical norms, with extra care given during scale-up or when handling solvents and mother liquors containing unreacted starting materials. Experience teaches that respect for basic safety avoids problems in both small and large labs.
The reach of this compound stretches past the synthetic lab. Pharmaceutical research keeps it in the toolbox for new bioactive scaffolds—those acetoacetamide moieties often serve as keys for enzyme inhibition or receptor blocking. Material scientists look to the bis-amide structure for cross-linking agents or as nodes in advanced polymer networks. Coordination chemists see opportunities for metal binding and sensing applications, using the dual functional groups to create rigid, predictable ligands. Some researchers even explore the pigment and dye space, taking advantage of the aromatic core and side chain reactivity for customized colorants or optical materials.
Chemists never stop poking at familiar compounds, trying new spins and seeing old problems with fresh perspective. The molecule’s popularity keeps rising, fueled by access to high-throughput screening and computational modeling. Medicinal chemists borrow from its structure to craft drug leads or design enzyme inhibitors. Others push into polymer science, aiming for selective membranes or robust frameworks. Researchers have also proposed using derivatives as intermediates in complex natural product syntheses, bridging gaps in synthetic routes that once seemed too difficult or inefficient.
Nobody wants nasty surprises from the chemicals on the bench. Toxicity information on N,N'-(1,4-Phenylene)bis(acetoacetamide) still develops, but early studies point toward low acute toxicity, typical for compounds without obvious reactive functional groups. Chronic studies are more limited, so conservative handling stays smart. Any molecule with aromatic amide content deserves careful evaluation over time due to rare but real risks of slow metabolic breakdown products in vivo. Larger studies, possibly with animal models and long-term assays, could help catch any unexpected hazards sooner rather than later. Having databases that aggregate up-to-date toxicity findings gives chemists and users a valuable line of defense.
Future work looks bright. This compound, with its unique blend of sites for reaction, offers a platform for advances in drug discovery, materials science, and complex organic synthesis. Improved synthesis will lower cost and increase availability, making it more attractive for interdisciplinary teams. Artificial intelligence and computational screening stand poised to identify unforeseen applications and predict valuable modifications. As research deepens, expect more collaborative projects linking fields as diverse as pharmacology, environmental chemistry, and renewable materials, driven by both creativity and pressing, real-world needs.
People often encounter a long chemical name and tune out, thinking it's only for academics or niche researchers. But some compounds, like N,N'-(1,4-Phenylene)bis(acetoacetamide), play an overlooked but significant role in research and industrial chemistry. Its structure holds clues to the types of reactions it enables and the applications it can support.
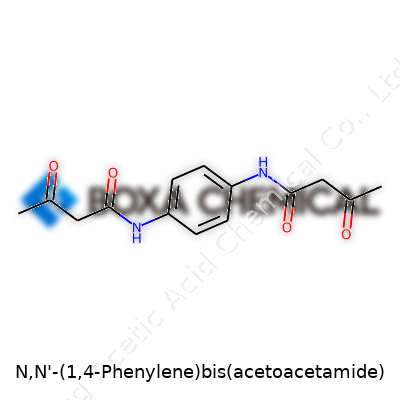
This compound brings together the acetoacetamide group with a benzene ring. Start with the 1,4-phenylene core, which comes from benzene—a well-known six-carbon ring with alternating double bonds. Instead of attaching random groups anywhere, in this molecule the modifications land at the para positions, which sit opposite each other around the ring. That symmetry influences both the chemical reactions it allows and the physical properties people care about in synthesis work.
On each of those positions, you find an acetoacetamide group. What this means in plain language: The benzene ring bonds to a nitrogen atom, and from there, each arm extends out to a carbonyl-carbon—the acetoacetyl group. The formula looks like this: C6H4(NHCOCH2COCH3)2. If you drew it, you would spot a central ring with two identical, branch-like arms going out at opposite sides, each built from an amide linked to a simple chain. The presence of two amide groups and two acetoacetyl components brings about hydrogen bonding and the ability to interact in interesting ways with metal ions or other molecules.
In the reality of the lab, researchers care about this kind of molecule for the doors it opens: From my time in synthesis labs, I've seen how bis-acetoacetamides find use as building blocks for making more complex chemicals. Their structure makes them candidates in areas like dye chemistry, pharmaceuticals, and polymer research, because those terminal amide groups and carbonyls react in predictable, controllable ways. The anchored benzene core helps stabilize the molecule, reducing unwanted side reactions.
The ability of this molecule to act as a chelating agent stands out. Chelating agents connect to metals through multiple points, and structures like this—with well-placed amide and carbonyl groups—easily form rings around metal ions. That has real-world importance in analytical chemistry, environmental cleanup (pulling metals from water), and in medical imaging.
One challenge I notice: Sourcing pure, consistent batches. Impurities can change reactivity, and even minor changes in the synthesis route affect outcomes. Methods for improving purity and greener synthesis routes are always under discussion in professional circles. Developing more sustainable ways to produce bis-acetoacetamides without harsh solvents or wasteful reagents would move the field forward. Open sharing of synthesis protocols among academic and industrial teams helps problem-solvers everywhere build off collective experience.
A deep understanding of core chemical structures—like N,N'-(1,4-Phenylene)bis(acetoacetamide)—gives researchers and innovators tools to craft better materials and tackle new challenges. Curiosity about chemical structure isn’t just academic; it shapes advances in medicine, environmental science, and production efficiency.
As someone who pays attention to chemistry inside and outside the lab, I’ve noticed that most folks barely notice the molecular building blocks shaping the products they use every day. Names like N,N'-(1,4-Phenylene)bis(acetoacetamide) rarely make it past technical circles, yet they do leave a visible mark in a range of industries. There’s a story behind this compound’s rise that reveals a lot about modern materials and the way we tackle real-world challenges.
No need to sugarcoat it—most people encounter the impact of this compound through products, not nomenclature. Chemists recognize N,N'-(1,4-Phenylene)bis(acetoacetamide) as a useful intermediate. It steps up during the manufacture of specialty polymers and resins. Think about how colorfast fabrics or durable coatings hold up against bleach, sweat, or sun. The answer often circles back to decisions made at the molecular level, far from the store shelf.
Its acetoacetamide groups serve a key role in creating cross-linked structures. This translates into stronger bonds within plastics or adhesives. For any industry looking for durability—be it automotive, electronics, or packaging—this feature tips the scales toward reliability. Growing up, I worked in a small electronics repair shop. I learned that even small tweaks in resin composition can shift a device from landfill-bound to long-lasting. Materials like this compound make those tweaks possible and affordable.
If you've ever wondered why some pigments hold their shade for years or why your favorite waterproof jacket hasn't faded, chemistry answers it clearly. N,N'-(1,4-Phenylene)bis(acetoacetamide) acts as a backbone for synthesizing stabilized dyes and pigments. This keeps color performance consistent in products from paints to textiles. The market favors solutions with staying power—nobody wants to repaint their living room every year or see a red shirt become salmon-pink after a few washes. With this compound’s input, pigments handle exposure better, enduring long sunbaths or harsh detergents.
The pharmaceutical industry also taps into its versatile backbone. While drug development sometimes feels like a lottery, robust intermediates matter. Laboratories use this molecule when designing new drugs, usually in the research phase. Solid foundations make every subsequent experiment less of a gamble. During one stint with a pharmaceutical start-up, our team hunted for intermediates that would lower costs without sacrificing purity. Intermediates with the structural consistency provided by this compound helped us bridge that gap, especially in early lead optimization.
No single ingredient solves every problem. Handling specialty chemicals brings environmental and safety challenges. I’ve seen what happens when teams skip risk assessment to cut costs. The result often means chemical waste that’s difficult to manage or unexpected hazards during scale-up. Regulatory expectations keep rising, especially around substances with persistence in the environment. Companies seeking long-term success run real-life safety drills, not just paperwork exercises. They choose greener synthesis routes where possible and plan for end-of-life recycling in finished materials.
There’s talk in research journals about bio-based alternatives and more sustainable variants. Efforts like these offer a way forward, helping industries keep pace with environmental goals. If your work touches materials science or product design, keeping an eye on these advances can pay off, saving resources and hassle down the line.
Understanding the chemistry behind N,N'-(1,4-Phenylene)bis(acetoacetamide) is not about memorizing another complex name—it’s about seeing the repeated impact of careful design. By following the path from molecule to market, we start to appreciate why attention to chemical detail shapes the quality of everyday life.
I can’t count how many times I’ve stood in a store aisle, squinting at a label, wondering what, exactly, I’m about to buy. Whether it’s a vitamin, cleaning powder, or even flour, one thing tends to stand out: everyone wants to know about purity and what the label really tells us. Transparency builds trust. When a manufacturer declares what percentage of a component you’re getting, that’s a statement of integrity. Sloppy or vague wording can leave consumers guessing—or worse, put health and businesses at risk.
For some people, this is a daily headache. Lab professionals check Certificates of Analysis line by line. Workers on production lines want to know that what’s in the shipment matches what their process needs. Customers notice too. If you ever had a batch of product come out wrong because of mystery contaminants or varying ingredient quality, you remember. Problems show up as recalls, complaints, and long days chasing down answers.
I once worked with a company that produced supplements. Ingredients shipped in from halfway around the world, each barrel with its own lab report. Trouble popped up when the percentage didn’t match what the supplier had claimed. Orders had to stop, formulas tweaked, timelines pushed back. In that moment, specifications weren’t just shelf labels. They ruled the entire flow of business, payroll, and trust with partners and customers.
Purity means more than a number on a spec sheet. In the world of food and pharmaceuticals, small mistakes in purity—say an unwanted heavy metal or a bit too much moisture—can cost lives, not just dollars. In the labs, a slight impurity might ruin weeks of experiments. Investors and regulators pay careful attention to these details because corners cut at this stage snowball into larger, costlier mistakes later on.
Data shows that good control over product specifications reduces recalls by as much as 50 percent. The U.S. Food and Drug Administration lists quality consistency among the top reasons for inspection failures. Independent testing helps, but overreliance on paperwork instead of robust in-house testing brings more risk.
Building a strong standard doesn’t start with a form or a stamp; it starts with honest sourcing, repeatable testing, and openness about a product’s real makeup. Testing every lot might raise costs, but that cost fades next to the price of bad publicity or lost market share. Trust doesn’t return easily once broken.
Technology also eases these challenges. Modern labs use spectrometry, chromatography, and other advanced tools to measure exact proportions and spot impurities at minuscule levels. Companies share these findings with the public, and that’s not just marketing spin—it’s proof. Tools like blockchain ledgers track authenticity from point of origin to end user, locking down tampering or substitution.
Knowledge about what you’re really getting gives power to everyone along the supply chain. From a parent choosing a snack for their child to a researcher running an experiment, nobody wants uncertainty. Clear details on purity and rigorous specifications carry more than technical weight—they send a message that the provider values safety, clarity, and respect for every dollar and every decision. That’s what moves the world forward, one honest label at a time.
Once you spend enough time in a laboratory or around industrial chemicals, you see how the small stuff matters just as much as the big reactions. Some compounds seem easy on paper but show their quirks once you get hands-on with tubs and jars. N,N'-(1,4-Phenylene)bis(acetoacetamide) fits that bill. This solid doesn’t get as much spotlight as acids or solvents, but it asks for a little respect in how you manage it.
It’s common to focus on reactivity or nice glossy product data, yet storage mistakes can cause headaches or even worse. Anyone who's opened a bottle to find sticky clumps or discoloration knows the pain. This isn’t unique to this compound, but with N,N'-(1,4-Phenylene)bis(acetoacetamide), humidity and stray exposure can make a difference. Keep it away from moisture and light. A dry place, tightly sealed, always wins. Not everybody has a climate-controlled storeroom, but placing bottles in a cool, shaded, reliable spot and checking seals goes a long way.
You don’t often see this chemical discussed outside of specialty labs or manufacturers, but I’ve seen enough variability from warehouse mixing and neglect. One batch looks fine, the next arrives caked or changed. This isn’t about fancy equipment; even a clear container and a silica packet can help for most labs. Simple tricks keep it stable, and those few extra minutes of care can be the difference between usable material and something fit for a waste drum.
Some folks ask for the datasheet and stop there. Experience says gloves and goggles are not optional here. Even a careful worker sometimes gets caught out by fine powders or unexpected spills. Once I saw a spill go airborne, and it took hours to clean up every trace. Always use it in a spot with good airflow, and if possible, a fume hood. Your nose and skin both prefer that.
There’s nothing noble in cutting corners. Wash up afterward, and make sure benches stay tidy. Tossing containers into a crowded drawer isn’t smart—chemicals deserve a proper shelf, labeled, and out of reach from food or careless break-ins. These aren’t fancy moves, but too often I've seen people let their guard down by treating everything like table salt.
Mistakes become stories that circulate among scientists and warehouse teams alike. More than once, a misguided shortcut forced a whole team to refinish surfaces or dump supplies. One careless spill or humid storeroom leads to ruined stock and expensive cleanup. Over time, good habits spread—the most respected workers know procedures by heart and reinforce them with new hires. Training isn’t just paperwork. Watching someone who’s careful rubs off on the team.
Consistent practice, along with a dash of pride in a clean bench and a well-sealed container, matters. This chemical may not win awards for drama, but it deserves smart handling. That sort of respect reduces risk and saves money in the long run. The more people own the safety culture, the fewer accidents everyone faces.Staying safe around chemicals isn’t just about following rules on paper. It takes a nose for detail and a measure of experience, especially with complex organic substances like N,N'-(1,4-Phenylene)bis(acetoacetamide). This mouthful of a compound rarely turns up in news headlines, but it does pop up in chemical synthesis and research labs around the world. Like many who’ve clocked countless hours in a lab coat, I’ve learned that chemicals with long names can invite short shrift—until something goes wrong. That’s why the big question isn’t only whether it’s useful, but whether it’s safe to use.
Few chemicals spark as much confusion in safety circles as lesser-known intermediates. I’ve looked, and the usual go-to resources like PubChem and ChemSpider list structural details for N,N'-(1,4-Phenylene)bis(acetoacetamide), but they skip on deeper toxicity insights or long-term exposure effects. Chemists with a practical streak tend to look up material safety data sheets (MSDS), but searching for this compound usually brings up more blanks than answers. This isn’t just a gap in a database—it’s a real-life problem once the compound leaves the bottle and enters a busy lab.
There’s a broad rule in chemistry: if you don’t know a chemical’s hazards, treat it with more care than the ones you do. I’ve always respected that policy, especially for compounds that could be derivatives of acetoacetamide, which sometimes act as irritants, even if not classified as highly toxic. Reading and handling an unknown like this feels a bit like walking down unlit stairs—you take it slow and never assume safety. Gloves, goggles, and good ventilation become routine, but they only go so far without solid information.
Information blindness can lead to unexpected accidents. I remember an acquaintance who handled a little-known organic molecule thinking it was “probably fine.” Later, they found out it left their skin irritated and their lungs burning, though nobody could point to solid toxicology. That uncertainty is the real enemy in chemistry, not just the chance of an explosive reaction. Just because no stories exist about this compound’s hazards doesn’t mean they don’t sit beneath the surface.
Researchers can push for more frequent and thorough chemical safety assessments, especially for compounds used in academic and industrial labs. Regulatory bodies like OSHA or ECHA in Europe keep massive databases—expanding them for lesser-known molecules should be part of future policy. Professional networks, both online and in real-world lab meetings, can crowdsource early warning signs or best practices, sharing them before journals catch up. Taking time to publish even minor toxicity findings, or flag unexplored risks, helps the next person who encounters this compound do so with eyes open.
Until the gap closes, working with the unknown calls for old-fashioned caution. Relying on tested personal protective equipment remains crucial, and seeking advice from experienced colleagues often saves time and trouble. Trusting instincts—if a compound looks strange or acts unpredictably—can keep problems from spreading beyond the lab bench. This kind of caution doesn’t slow down progress; it protects those doing the work.
No one wants to wind up as the cautionary tale in their department. Keeping an eye on compounds like N,N'-(1,4-Phenylene)bis(acetoacetamide) means acting before problems appear in the literature or the press. Open conversation, sharing real stories, and insisting on more data lay the groundwork for safer science. Respecting the unknown in chemistry has always been the smarter bet.